Search
Thermo Scientific Chemicals
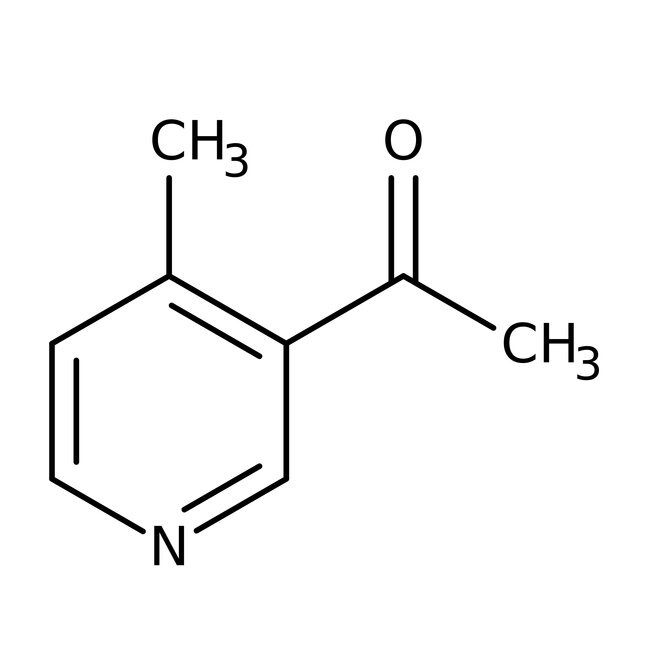
3-Acetyl-4-methylpyridine, 96%
CAS: 51227-30-6 | C8H9NO | 135.166 g/mol
Catalog number H33380.03
also known as H33380-03
Price (USD)
61.65
Special offer
Online exclusive
Ends: 30-Jun-2026
72.70Save 11.05 (15%)
Each
Quantity:
1 g
Price (USD)
61.65
Special offer
Online exclusive
Ends: 30-Jun-2026
72.70Save 11.05 (15%)
Each
Chemical Identifiers
CAS51227-30-6
IUPAC Name1-(4-methylpyridin-3-yl)ethan-1-one
Molecular FormulaC8H9NO
InChI KeyLCEINQHGZAFADC-UHFFFAOYSA-N
SMILESCC(=O)C1=C(C)C=CN=C1
View more
Specifications Specification Sheet
Specification Sheet
FormLiquid
Appearance (Color)Clear colorless to pale yellow to pale brown
Assay (GC)≥95.0%
Refractive Index1.5265-1.5325 @ 20?C
It is used as pharmaceutical intermediate. Benzoylthiophenes are allosteric enhancers (AE) of agonist activity at the A1 adenosine receptor.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
It is used as pharmaceutical intermediate. Benzoylthiophenes are allosteric enhancers (AE) of agonist activity at the A1 adenosine receptor.
Solubility
Sparingly soluble in water.(0.26 g/L) (25°C),
Notes
Store in cool dry place. Ensure proper ventilation. Incompatible with oxidizing agents.
It is used as pharmaceutical intermediate. Benzoylthiophenes are allosteric enhancers (AE) of agonist activity at the A1 adenosine receptor.
Solubility
Sparingly soluble in water.(0.26 g/L) (25°C),
Notes
Store in cool dry place. Ensure proper ventilation. Incompatible with oxidizing agents.
RUO – Research Use Only
General References:
- C. Elisabet Tranberg.; Andrea Zickgraf.; Brian N. Giunta.; Henning Luetjens.; Heidi Figler.; Lauren J. Murphree.; Ruediger Falke.; Holger Fleischer.; Joel Linden.; Peter J. Scammells.; Ray. A. Olsson. 2-Amino-3-aroyl-4,5-alkylthiophenes: Agonist Allosteric Enhancers at Human A1 Adenosine Receptors.J. Med. Chem. 2002, 45 (2),382-389 .
- Robert H. Dodd.; Catherine Ouannes.; Malka Robert-Gero.; Pierre Potier. Hybrid molecules: growth inhibition of Leishmania donovani promastigotes by thiosemicarbazones of 3-carboxy-.beta.-carbolines.J. Med. Chem. 1989, 32 (6),1272-1276 .