Search
Thermo Scientific Chemicals
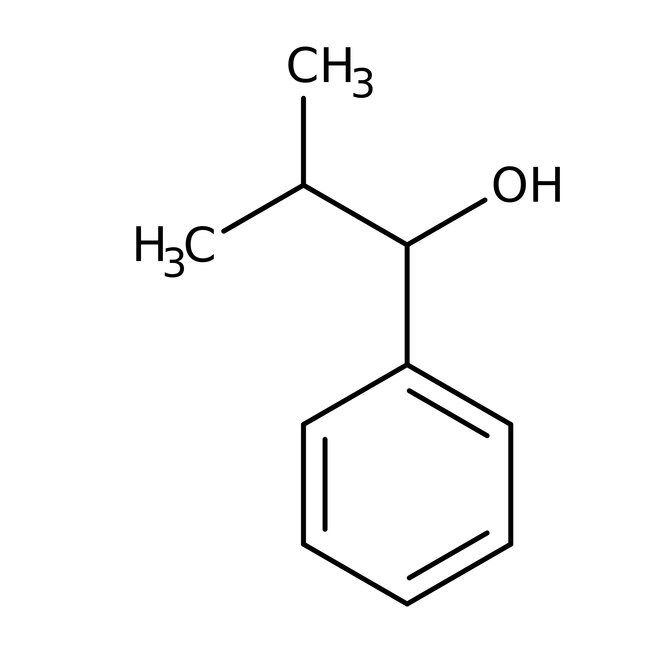
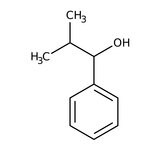
2-Methyl-1-phenyl-1-propanol, 98%
CAS: 611-69-8 | C10H14O | 150.221 g/mol
| Catalog Number | Quantity |
|---|---|
| B20729.06 also known as B20729-06 | 5 g |
Chemical Identifiers
CAS611-69-8
IUPAC Name2-methyl-1-phenylpropan-1-ol
Molecular FormulaC10H14O
InChI KeyGMDYDZMQHRTHJA-UHFFFAOYNA-N
SMILESCC(C)C(O)C1=CC=CC=C1
View more
Specifications Specification Sheet
Specification Sheet
Assay (GC)≥97.5%
Appearance (Color)Clear colorless
Refractive Index1.5115-1.5155 @ 20?C
FormLiquid
It is used in the large-scale preparation of (R)-and (S)-1-(2, 4, 6-triisopropylphenyl) ethanol and also in the baker's yeast mediated asymmetric reduction of cinnamaldehyde derivatives.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
It is used in the large-scale preparation of (R)-and (S)-1-(2, 4, 6-triisopropylphenyl) ethanol and also in the baker′s yeast mediated asymmetric reduction of cinnamaldehyde derivatives.
Solubility
Not miscible or difficult to mix in water.
Notes
Store away from oxidizing agents. Keep the container tightly closed and place it in a cool, dry and well ventilated condition.
It is used in the large-scale preparation of (R)-and (S)-1-(2, 4, 6-triisopropylphenyl) ethanol and also in the baker′s yeast mediated asymmetric reduction of cinnamaldehyde derivatives.
Solubility
Not miscible or difficult to mix in water.
Notes
Store away from oxidizing agents. Keep the container tightly closed and place it in a cool, dry and well ventilated condition.
RUO – Research Use Only
General References:
- Philippe Delair, et al. Efficient, large-scale preparation of (R)- and (S)-1-(2,4,6-triisopropylphenyl)ethanol, versatile chiral auxiliary for cyclopentenone, γ-butyrolactone, and γ-butyrolactam synthesis.Tetrahedron: Asymmetry.,1996,7(9), 2707-2710.
- H. N. Stephens. Oxidation In The Benzene Series By Gaseous Oxygen. Iv. Mechanism Of The Slow Oxidation Of Saturated Hydrocarbons.J. Am. Chem. Soc.1928,50(9), 2523-2529.