Search
Thermo Scientific Chemicals
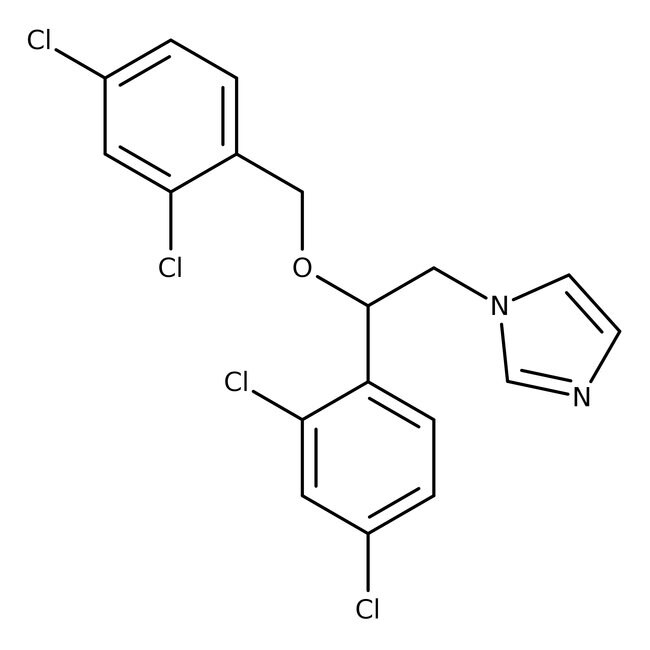
Miconazole, 97%
Inhibits cytochrome P450-dependent 14-alpha-demethylase | CAS: 22916-47-8 | C18H14Cl4N2O | 416.12 g/mol
Chemical Identifiers
CAS22916-47-8
IUPAC Name1-[2-(2,4-dichlorophenyl)-2-[(2,4-dichlorophenyl)methoxy]ethyl]-1H-imidazole
Molecular FormulaC18H14Cl4N2O
InChI KeyBYBLEWFAAKGYCD-UHFFFAOYNA-N
SMILESClC1=CC=C(COC(CN2C=CN=C2)C2=CC=C(Cl)C=C2Cl)C(Cl)=C1
View more
Specifications Specification Sheet
Specification Sheet
Melting Point75.0-82.0°C
Appearance (Color)White
FormPowder
Assay from Supplier's CofA≥96.0% (HPLC)
Miconazole, is used as an antifungal inhibitor of aromatase. Miconazole has been shown to promote remyelination of neurons in chronic progressive multiple sclerosis mouse models. Miconazole is mainly used externally for the treatment of athlete's foot, ringworm, and jock itch. Internal application is used for oral or vaginal thrush (yeast infection). The oral gel may also be used for the lip disorder angular cheilitis. It is also used in photography.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Solubility
Freely soluble in alcohols or acetone. Also soluble in DMF or chloroform. Insoluble in water
Notes
Protect from light. Store in well-closed containers.
Freely soluble in alcohols or acetone. Also soluble in DMF or chloroform. Insoluble in water
Notes
Protect from light. Store in well-closed containers.
RUO – Research Use Only
General References:
- White TC.; Marr KA.; Bowden RA. Clinical, cellular, and molecular factors that contribute to antifungal drug resistance. Clin Microbiol Rev. 1998, 11,(2), 382-402.
- Ghannoum MA.; Rice LB. Antifungal agents: mode of action, mechanisms of resistance, and correlation of these mechanisms with bacterial resistance. Clin Microbiol Rev. 1999, 12,(4), 501-17.