Search
Thermo Scientific Chemicals
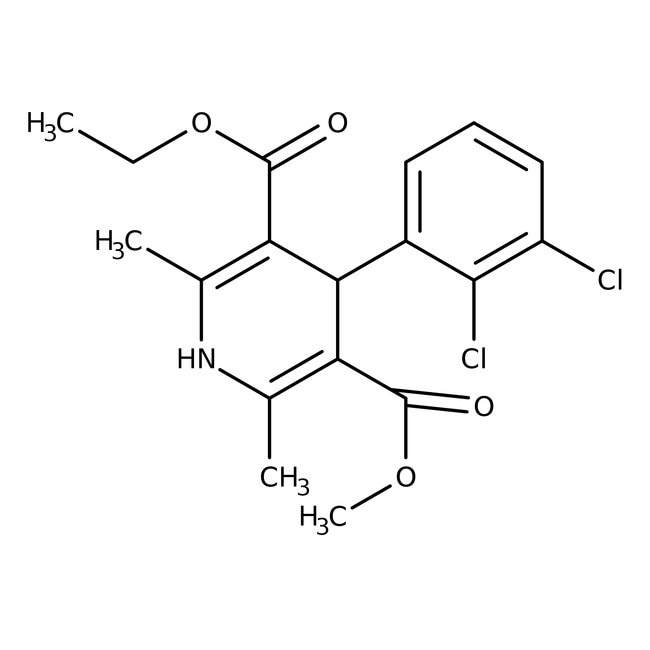
Felodipine
A dihydropyridine calcium channel blocker | CAS: 72509-76-3 | C18H19Cl2NO4 | 384.253 g/mol
| Catalog Number | Quantity |
|---|---|
| J61195.MC also known as J61195-MC | 100 mg |
Catalog number J61195.MC
also known as J61195-MC
Price (CNY)
-
Quantity:
100 mg
Chemical Identifiers
CAS72509-76-3
IUPAC Name3-ethyl 5-methyl 4-(2,3-dichlorophenyl)-2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate
Molecular FormulaC18H19Cl2NO4
InChI KeyRZTAMFZIAATZDJ-UHFFFAOYNA-N
SMILESCCOC(=O)C1=C(C)NC(C)=C(C1C1=CC=CC(Cl)=C1Cl)C(=O)OC
View more
Specifications Specification Sheet
Specification Sheet
Purity≥95.0%
Mass SpecConforms
FormPowder or crystals or crystalline powder
Appearance (Color)Yellow to yellow-green
Felodipine is used as a dihydropyridine calcium channel blocker. It displays high vascular selectivity; lowers arterial blood pressure without altering cardiac contractility. Antihypertensive.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Felodipine is used as a dihydropyridine calcium channel blocker. It displays high vascular selectivity; lowers arterial blood pressure without altering cardiac contractility. Antihypertensive.
Solubility
Soluble in DMSO. Insoluble in water
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Keep away from strong oxidizing agents.
Felodipine is used as a dihydropyridine calcium channel blocker. It displays high vascular selectivity; lowers arterial blood pressure without altering cardiac contractility. Antihypertensive.
Solubility
Soluble in DMSO. Insoluble in water
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Keep away from strong oxidizing agents.
RUO – Research Use Only
General References:
- Ljung. Vascular selectivity of felodipine: experimental pharmacology. J.Cardiovasc.Pharmacol. 1990, 15,(4), S11-6.
- Furukawa et al. Selectivities of dihydropyridine derivatives in blocking Ca2+ channel subtypes expressed inXenopus oocytes. J.Pharmacol.Exp.Ther. 1999, 291,(2), 464-73.