Interested in HPLC method modernization and compliance? I answer the most common customer questions and show how optimization dramatically reduces analysis time while boosting efficiency and maintaining regulatory compliance.

Across all my visits to pharmaceutical manufacturing sites, there’s one question customers consistently ask: How can laboratories increase productivity without compromising regulatory compliance?
From quality control to research and development, teams face the same reality. Analytical demands keep growing, the number of samples continues to rise, and regulatory expectations are evolving at the same time.
A client in a pharma QC lab had a seemingly simple question for me: Which Chromatographic parameters are adjustable without triggering method revalidation?
Behind these questions lies a broader challenge — cutting solvent use and run time on existing methods while remaining fully aligned with regulatory submissions already in place.
Below, I’ll explore these parameters and the regulatory frameworks that make such optimization possible.
UHPLC modernization and workflow transformation
Method validation is the cornerstone of reliable pharmaceutical analysis, making any decision to optimize a routine method particularly sensitive.
Beyond evolving regulatory expectations and efficiency goals, validation is also essential to mitigate the risk of out-of-specification results that could directly impact batch release and trigger costly investigations.
Yet beyond the constraints, optimization is a powerful value-creation lever. While it does require time and resources, a structured approach — aligned with current regulatory frameworks and supported by modern analytical technologies — can deliver substantial benefits.
A case study conducted at the Opella site (formerly Sanofi) in Compiègne, France, provides a great example, demonstrating how the modernization of HPLC methods to UHPLC sustainably transforms routine analytical workflows.
Key takeaways:
- Unlock productivity without compromising compliance through UHPLC method modernization
- Leverage regulator-enabled frameworks that make method modernization no longer optional, but supported
- Realize measurable cost savings — up to $7,070 per year, as demonstrated by Opella with a single method

Opella case study: How regulations are driving method modernization
Method optimization and revalidation are no longer isolated initiatives or internal laboratory trade-offs. They are part of a broader regulatory shift that now encourages a more structured and proactive approach to analytical method optimization throughout the entire method lifecycle.
In this context, the introduction of USP <1220> in 2022 marked a key milestone, laying the foundation for an analytical lifecycle management strategy built on method understanding, risk-based decision-making, and continuous improvement — rather than a static view of compliance.
With the recent adoption of the ICH Q14 guideline on analytical procedure development, regulatory authorities have formally acknowledged the value of continuous method improvement, provided changes are scientifically justified and appropriately documented. Rather than locking analytical methods into their initial state, this framework actively supports instrument and method modernization, including transitions to higher-performance technologies such as UHPLC, while maintaining a high level of regulatory control.
In parallel, updates to USP <621> introduce greater flexibility for chromatographic methods, allowing certain adjustments — such as column dimensions, flow rate, or gradient parameters — without requiring full method revalidation.
This regulatory evolution provides laboratories with a practical lever to optimize existing methods, shortens analysis times, and reduces solvent consumption, all while remaining fully compliant with pharmaceutical requirements.
Taken together, ICH Q14, USP <621>and the ICH Q2 (R2) fundamentally reshape how laboratories can approach analytical modernization. Far from adding complexity, these guidelines turn method optimization into a strategic opportunity — one that aligns operational performance, regulatory compliance, and economic objectives.
By emphasizing deeper understanding of critical method parameters, performance-based validation, and structured lifecycle management, ICH Q14 and ICH Q2 (R2) clearly connect method development, validation, and continuous improvement.
Combined with the flexibility introduced by USP <621>, this regulatory landscape provides laboratories with a clearer and more confident path to modernizing LC methods and transitioning to UHPLC platforms — an approach illustrated by Opella.
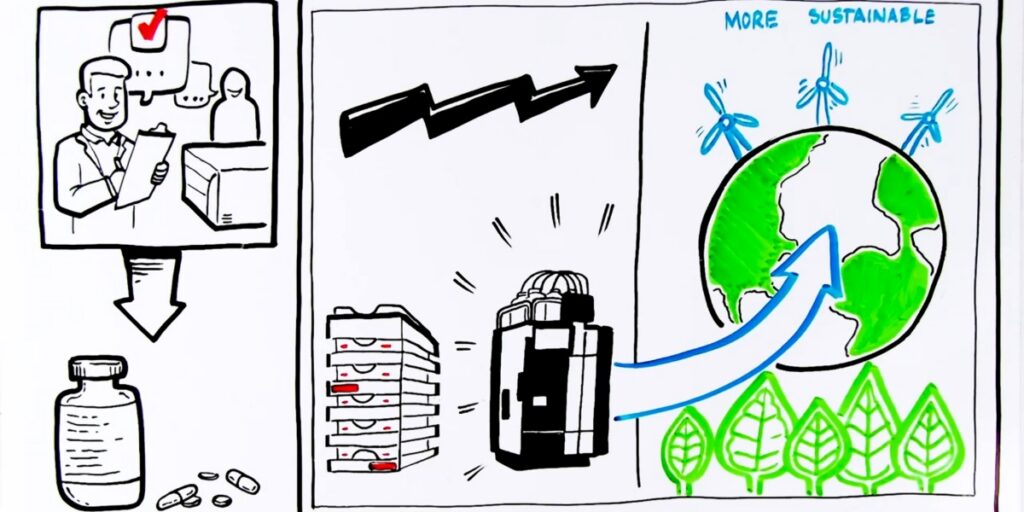
HPLC method modernization reduced run times by 90% at Opella
At the Opella Site in Compiègne, analytical method modernization proved to be far more than a technical upgrade. By taking a structured approach and modernizing routine LC methods to a Vanquish Flex UHPLC platform, the teams unlocked a new level of efficiency.
Two aging LC methods consolidated into a single UHPLC method. Alongside improved chromatographic performance, the teams strategically redesigned sample preparation, further contributing to time and cost savings, with a clear objective: increase productivity for a method used across two manufacturing sites, supporting approximately 300 lots per year distributed worldwide.
The impact was immediate. Analyses that once took nearly an hour are now completed in just minutes, with run times reduced by more than 90%. This modernization dramatically increases analytical capacity, significantly reduces solvent consumption, and simplifies day-to-day laboratory operations. Applied across two manufacturing sites, these gains saved 20 working days per year, creating additional capacity for higher-value analytical work while enhancing cost efficiency, safety, and sustainability.
This example demonstrates how UHPLC modernization, when supported by full method revalidation and updates to manufacturing authorization documentation, can turn routine LC workflows into a lasting operational advantage.
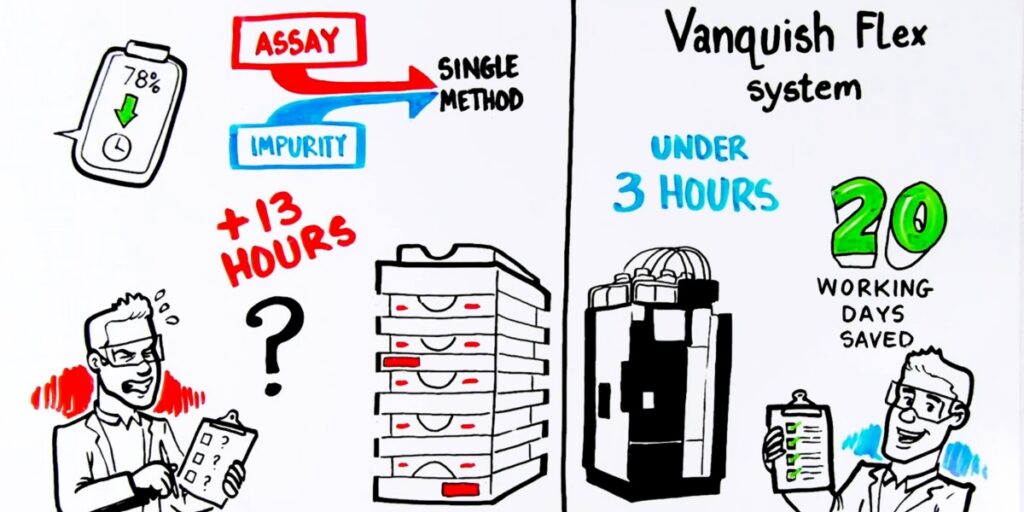
Steps your lab can take to modernize legacy methods
Modern analytical methods aren’t defined solely by performance. Today, a “modern” method is faster, greener, and less error prone. It’s designed to reduce analysis time, solvent consumption, and operational risk while remaining fully compliant.
The first step toward modernization is prioritization. Laboratories should start by identifying methods with the greatest impact on daily operations. These are typically high-volume methods used for routine testing, legacy methods developed 20 years ago or more, or methods that frequently generate deviations, investigations, or operator errors.
From there, two strategic paths typically emerge, depending on the scope of change and regulatory requirements.
Path 1: Optimize within USP <621> — fast and low risk
The simplest and fastest approach is to optimize methods within the flexibility allowed by USP <621>.
Laboratories can often adjust column dimensions, flow rate, gradient parameters, or injection volume without full revalidation or updates to regulatory authorizations, provided they demonstrate method performance equivalence.
In practice, this type of optimization was complete within a few weeks, delivering measurable gains in throughput and solvent reduction.
Path 2: Full method modernization — higher effort, greater impact
For more substantial improvements, a broader modernization strategy may be required, as illustrated by the approach taken at Opella. This path involves redesigning the method, upgrading instrumentation, and fully validating the new approach. While more resource-intensive, it unlocks the highest level of performance improvement.
Based on Opella’s experience, a typical timeline includes:
- Method development (1-2 months)
- Method validation (2-3 months)
- Regulatory dossier submission and approval (3-6 months)
While this global approach requires months to complete, the long-term benefits in productivity, robustness, sustainability, and operational confidence are well suited for high-impact, high-volume methods. By choosing the right path for each method, laboratories can modernize progressively, balancing quick wins with longer-term strategic transformations.

How Opella transformed routine LC workflows, reduced run times by over 90%, and unlocked measurable productivity and sustainability gains
Beyond the measurable productivity gains highlighted above, Opella also points to several practical advantages of the Thermo Scientific Vanquish Flex UHPLC System that directly support method optimization and day-to-day laboratory efficiency.
According to the analytical development team:
- The intuitive flow path design makes the system easy to understand and quick to adopt, reducing training time and simplifying daily operations.
- Integration and qualification using third-party software was smooth and straightforward — an important factor in regulated AD and QC laboratories.
- From a usability standpoint, Thermo Scientific Viper Fingertight Fittings eliminate the need for tools during column installation, enabling fast, reliable, and leak-free connections.
- Improved data reproducibility, with relative standard deviations below 1%, strengthens confidence in analytical results, while the absence of system suitability failures since adoption underscore the robustness of the platform in daily operation.
- The large autosampler capacity — up to 216 × 2 mL vials — allows complete dissolution profiles or high-throughput batch testing runs on a single system. For labs needing to scale throughput even further, the new Thermo Scientific Vanquish Sample Hub, accommodates up to 486 additional 2 mL vials, supporting extended, unattended operation for large analytical series.
Read the full case study today to explore the complete results, and lessons learned.

Xavier Lelièvre, now head of quality at Opella Compiègne recently notes that “by dramatically reducing analytical time and minimizing solvent use, the Thermo Scientific Vanquish Flex UHPLC sets a new standard for efficiency, sustainability and safety in the lab.”
Frequently asked questions
Yes. Vanquish systems can be operated with Thermo Scientific Chromeleon CDS as well as third-party systems, making integration into existing regulated workflow straightforward.
Yes. While the examples come from a regulated environment, the core benefits — faster analyses, lower solvent use, and simpler workflows — are applicable to all laboratory types (QC, AD, R&D) across industries (food, environmental, cosmetics etc.)
Not necessarily. Frameworks such as ICH Q14, ICH Q2 (R2), and updates to USP allow certain method adjustments without full revalidation, provided changes are scientifically justified and properly documented.
USP allows defined adjustments to parameters such as column dimensions, flow rate, gradient conditions, or injection volume, if method performance equivalence is demonstrated.
Visit us on LinkedIn: #UHPLC #hplc