Search
Invitrogen
Carboxypeptidase A1/A2/B Recombinant Rabbit Monoclonal Antibody (11A12)
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
图: 1 / 2
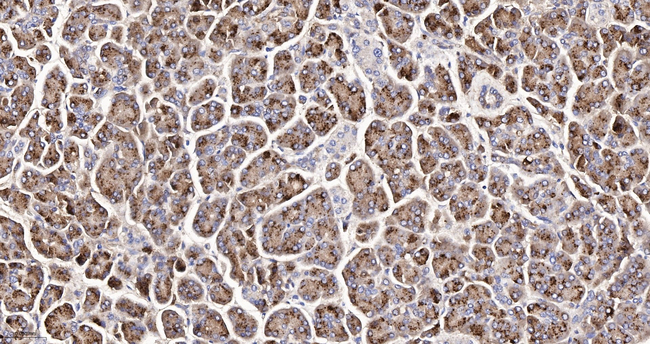
Carboxypeptidase A1/A2/B Antibody (MA573085) in IHC (P)

产品信息
MA573085
种属反应
宿主/亚型
Expression System
分类
类型
克隆号
抗原
偶联物
形式
浓度
规格
纯化类型
保存液
内含物
保存条件
运输条件
靶标信息
Carboxypeptidases are pancreatic proteases that digest proteins by cleaving amino acids from the carboxyl (C-terminal) end of peptide chains, with Carboxypeptidase A1 (CPA1), A2 (CPA2), and B (CPB) playing central roles in protein metabolism. CPA1, encoded by the CPA1 gene, is a zinc-dependent metalloenzyme that preferentially removes aromatic or branched-chain amino acids; it is synthesized in the pancreas as the inactive zymogen proCPA1 and activated by trypsin in the small intestine. CPA2, encoded by CPA2, is also a zinc-dependent metalloenzyme produced as an inactive proenzyme and activated by intestinal proteases; while it shares significant homology with CPA1, it differs in substrate specificity and regulation, removing aromatic or aliphatic amino acids from peptide C-termini. In contrast, CPB, encoded by CPB1, has distinct substrate specificity, primarily cleaving basic amino acids such as arginine and lysine from the C-terminus of peptides; like CPA1 and CPA2, it is synthesized in the pancreas as an inactive zymogen and activated by trypsin in the small intestine, where it contributes to the final stages of protein digestion and broader physiological regulation.
仅用于科研。不用于诊断过程。未经明确授权不得转售。