Search
Agrisera
HemC/Porphobilinogen-Deaminase (PBG) Polyclonal Antibody
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
图: 1 / 1
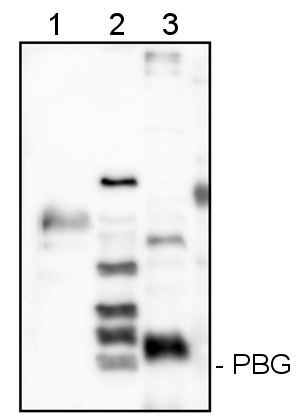
HemC/Porphobilinogen-Deaminase (PBG) Antibody (AS08-375) in WB
产品信息
AS08-375
种属反应
宿主/亚型
分类
类型
抗原
偶联物
形式
保存液
内含物
保存条件
运输条件
产品详细信息
Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube.
For reconstitution add 200 µL of sterile water.
Specific Species Reactivity: Thermosynechococcus elongatus
靶标信息
HemC encodes porphobilinogen deaminase (hydroxymethylbilane synthase), a soluble, cytosolic enzyme of the heme biosynthetic pathway that operates on tetrapyrrole intermediates rather than membranes or organelles. In bacteria, hemC is typically a single-copy locus within the core heme biosynthesis gene set and produces a monomeric enzyme of roughly 30-40 kDa that adopts an alpha/beta fold with a deep, positively charged active-site cleft. The enzyme contains an essential, covalently bound dipyrromethane cofactor attached to a conserved cysteine residue; this cofactor serves as a priming "handle" for stepwise chain elongation. Functionally, HemC catalyzes the sequential condensation (tetrapolymerization) of four porphobilinogen molecules to form the linear tetrapyrrole hydroxymethylbilane (also termed preuroporphyrinogen), providing the immediate substrate for uroporphyrinogen III formation in the next step of the pathway and thereby enabling downstream synthesis of heme and other biologically important tetrapyrroles.
仅用于科研。不用于诊断过程。未经明确授权不得转售。
篇参考文献 (0)
生物信息学
蛋白别名: hydroxymethylbilane synthase; porphobilinogen ammonia-lyase (polymerizing); porphobilinogen deaminase; pre-uroporphyrinogen synthase; uroporphyrinogen I synthase; uroporphyrinogen I synthetase