Search
GenScript
Durvalumab Monoclonal Antibody (4B5)
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
图: 1 / 2
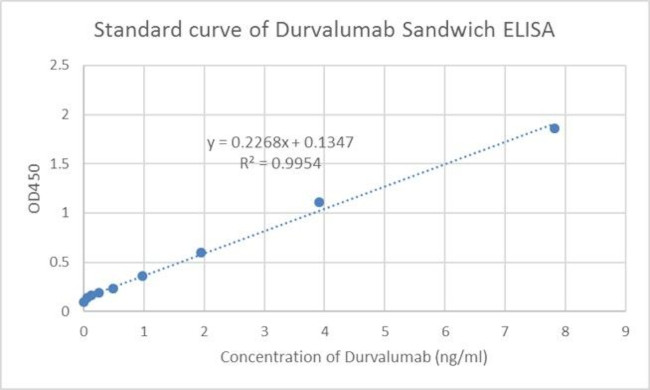
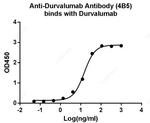
Durvalumab Antibody (A02030-40) in ELISA

产品信息
A02030-40
宿主/亚型
分类
类型
克隆号
抗原
偶联物
形式
规格
保存条件
运输条件
产品详细信息
Reconstitute the lyophilized antibody with deionized water (or equivalent) to a final concentration of 0.5 mg/ml.
Durvalumab with trade name Imfinzi, is an FDA-approved anti-cancer drug. It is known as a checkpoint inhibitor drug, developed by Medimmune/AstraZeneca. Durvalumab is a human IgG1kappa monoclonal antibody. It blocks the interaction of programmed cell death ligand 1 (PD-L1) with the PD-1 (CD279).Anti-Durvalumab Antibody (4B5), mAb, Mouse is produced from a hybridoma resulting from the fusion of partner and B-lymphocytes obtained from a mouse immunized with Durvalumab.
靶标信息
Programmed death receptor ligand 1 (PD-L1, also called B7-H1) is a recently described B7 family member. To date, one specific receptor has been identified that can be ligated by PD-L1. This receptor, programmed death receptor 1 (PD-1), has been shown to negatively regulate T-cell receptor (TCR) signaling. Upon ligating its receptor, PD-L1 has been reported to decrease TCR-mediated proliferation and cytokine production. PD-L1 expression was found to be abundant on many murine and human cancers and could be further up-regulated upon IFN-gamma stimulation. Thus, PD-L1 might play an important role in tumor immune evasion.
仅用于科研。不用于诊断过程。未经明确授权不得转售。