Search
GenScript
Presenilin 1 Polyclonal Antibody
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
图: 1 / 1
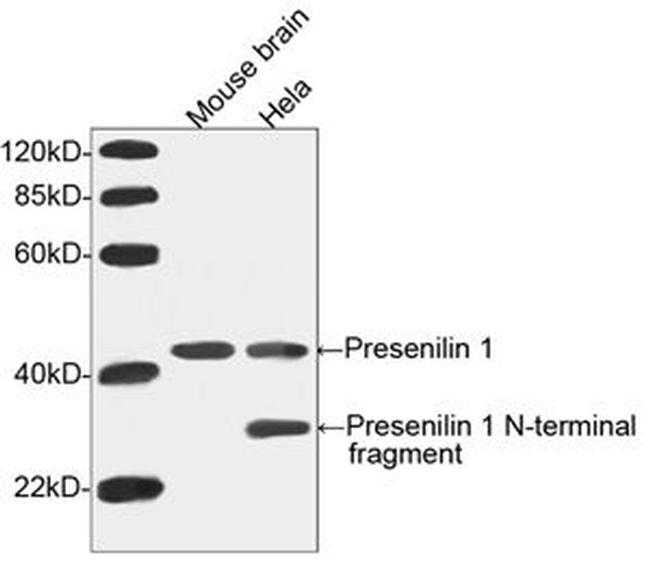
Presenilin 1 Antibody (A00881-40) in WB
产品信息
A00881-40
宿主/亚型
分类
类型
抗原
偶联物
形式
浓度
规格
保存条件
运输条件
产品详细信息
Presenilin 1 (PS1) and presenilin 2 (PS2) are catalytic subunits of the gamma-secretase complex, an endoprotease complex that catalyzes the intramembrane cleavage of integral membrane proteins such as Notch receptors and APP (beta-amyloid precursor protein). The other members of the gamma-secretase complex are required to have protease activity. PS1 and PS2 may play a role in intracellular signaling and gene expression or in linking chromatin to the nuclear membrane. It regulates epithelial-cadherin function. Three causative genes have been identified that when mutated lead to presenile Alzheimer's disease: APP (amyloid precursor protein gene), PSEN1, and PSEN2. The presenilin 1 gene has recently been discovered on chromosome 14 and is responsible for an early onset form of Alzheimer's disease that accounts for 70-80% of the inherited forms of Alzheimer's. It is a membrane protein that has a region where at least seven mutations have been identified in family members with Alzheimer's disease. Endogenous presenilin mainly exists in a heterodimeric complex formed from the endoproteolytically processed amino-terminal (28 kD) and carboxy-terminal (&(8)776;17 kD) fragments.GenScript Rabbit Anti-Presenilin 1 Polyclonal Antibody is developed in rabbit hosts using a synthetic peptide, TVRSQNDNRERQEHNDRRSLGHPEPL, corresponding to amino acids 25-50 of the N-terminus of human presenilin 1.Rabbit Anti-Presenilin 1 Polyclonal Antibody is highly purified from rabbit antiserum by immunoaffinity chromatography and is supplied as a 40 μg aliquot at a concentration of 0.5 mg/ml.
靶标信息
Presenilin1 was initially identified a marker of susceptibility to early-onset Alzheimer's disease. In addition to PEN2, nicastrin and APH-1, Presenilin1 forms the -gamma-secretase protein complex, a membrane-bound aspartyl protease that can cleave certain proteins at peptide bonds buried within the hydrophobic environment of the lipid bilayer. This cleavage is responsible for a key step in signaling from several cell-surface receptors and is thought to be required for the generation of the neurotoxic amyloid peptides that are central to the pathogenesis of Alzheimer's disease. Like the tumor necrosis factor-alpha-converting enzyme (TACE) and the beta-site cleavage enzyme (BACE) protease families, -gamma-secretase will cleave the amyloid precursor protein (APP), but within the intramembrane region of APP, resulting in either the non-toxic p3 (from the alpha and -gamma cleavage site) or the toxic Abeta amyloid peptide (from the beta and -gamma cleavage site). It is thought that accumulation of the Abeta peptide is the precursor to Alzheimer's disease. Multiple isoforms of presenilin1 are known to exist.
仅用于科研。不用于诊断过程。未经明确授权不得转售。