Search
Invitrogen
CD106 (VCAM-1) Monoclonal Antibody (429), eBioscience™
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
图: 1 / 9
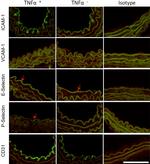
CD106 (VCAM-1) Antibody (14-1061-82) in ICC/IF, Flow

产品信息
14-1061-82
种属反应
已发表种属
宿主/亚型
分类
类型
克隆号
偶联物
形式
浓度
规格
纯化类型
保存液
内含物
保存条件
运输条件
RRID
产品详细信息
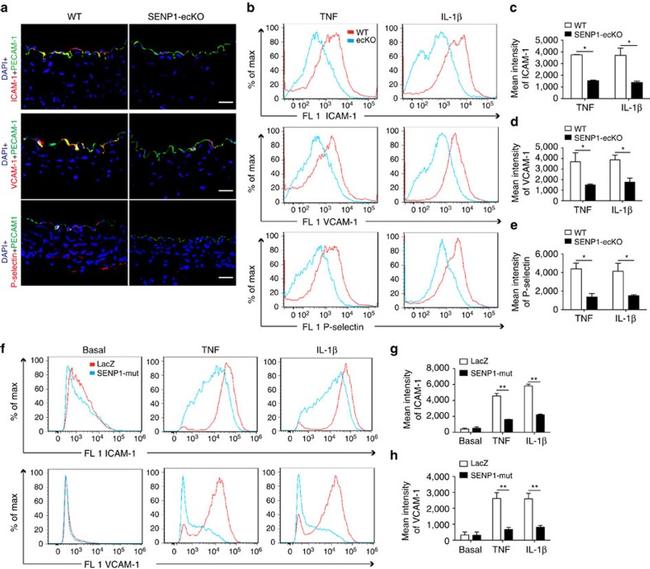
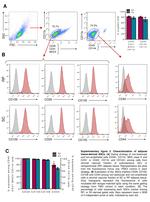
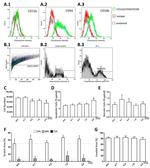
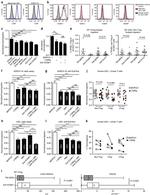
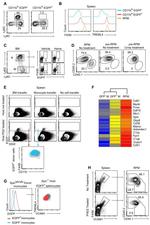
Description: The 429 monoclonal antibody reacts with mouse CD106 (Vascular Cell Adhesion Molecule-1, VCAM-1), a 110 kDa transmembrane glycoprotein expressed by myeloid lineage and bone marrow stromal cells. Endothelial cells constitutively express low levels of CD106 and upregulate it upon cytokine stimulation. CD106 binds to integrin alpha4beta1 (VLA-4, CD49d/CD29) and Integrin a4b7 (LPAM-1) and these interactions in the bone marrow and thymus are important for early lymphocyte and myeloid development. Cytokine-mediated upregulation of CD106 on endothelial cells suggests a role for this antigen in the inflammatory response.
Applications Reported: This 429 antibody has been reported for use in flow cytometric analysis, immunoprecipitation, and immunohistochemical staining of frozen tissue sections (IHC-F). It has also been reported in blocking of CD106 in functional studies. (Please use Functional Grade purified 429, Product # 16-1061, in functional assays).
Applications Tested: The 429 antibody has been tested by flow cytometric analysis of mouse bone marrow cells. This can be used at less than or equal to 0.25 µg per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest.
Purity: Greater than 90%, as determined by SDS-PAGE.
Aggregation: Less than 10%, as determined by HPLC.
Filtration: 0.2 µm post-manufacturing filtered.
靶标信息
VCAM-1 is a cell surface sialoglycoprotein expressed by cytokine activated endothelium. The protein has a number of functions including the regulation of leukocyte migration, leukocyte-endothelial cell adhesion and signal transduction and may play a role in a number of inflammatory diseases.
仅用于科研。不用于诊断过程。未经明确授权不得转售。
生物信息学
蛋白别名: DKFZp779G2333; INCAM100; MGC99561; sCD106; soluble CD106; soluble VCAM 1; sVCAM 1; V-CAM 1; vascular cell adhesion protein 1
基因别名: CD106; Vcam-1
Entrez Gene ID: (Mouse) 22329