Search
Invitrogen
CD8b Monoclonal Antibody (eBioH35-17.2 (H35-17.2)), eBioscience™
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promotions']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.viewpromo']}}
{{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.promocode']}}: {{promo.promoCode}} {{promo.promoTitle}} {{promo.promoDescription}}. {{$productOrderCtrl.translations['antibody.pdp.commerceCard.promotion.learnmore']}}
图: 1 / 9
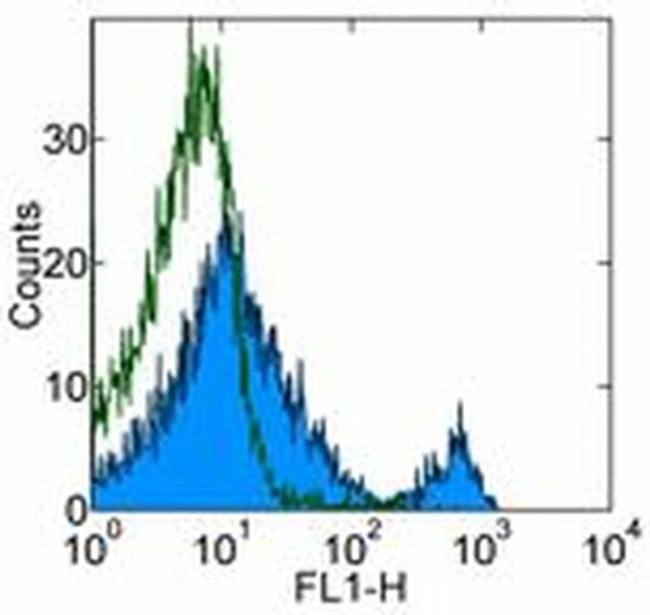
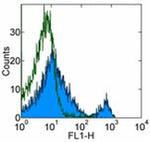
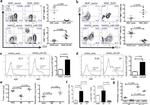
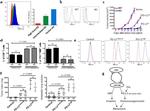
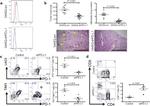
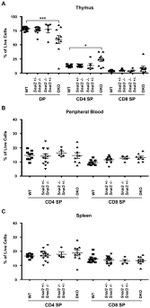
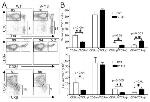
CD8b Antibody (14-0083-82) in Flow

产品信息
14-0083-82
种属反应
已发表种属
宿主/亚型
分类
类型
克隆号
偶联物
形式
浓度
规格
纯化类型
保存液
内含物
保存条件
运输条件
RRID
产品详细信息
Description: The eBioH35-17.2 monoclonal antibody reacts with the mouse CD8 beta molecule. The CD8 beta chain associates with the CD8 alpha chain to form the CD8 alpha/beta heterodimer expressed on the surface of a majority of thymocytes, and on peripheral cytotoxic alpha beta TCR T cells. CD8 binds to MHC class I and plays a role in T cell development and activation of mature T cells.
Applications Reported: This eBioH35-17.2 (H35-17.2) antibody has been reported for use in flow cytometric analysis, immunoprecipitation, and immunohistochemical staining of frozen tissue sections.
Applications Tested: This eBioH35-17.2 (H35-17.2) antibody has been tested by flow cytometric analysis of mouse splenocytes and thymocytes. This can be used at less than or equal to 0.5 µg per test. A test is defined as the amount (µg) of antibody that will stain a cell sample in a final volume of 100 µL. Cell number should be determined empirically but can range from 10^5 to 10^8 cells/test. It is recommended that the antibody be carefully titrated for optimal performance in the assay of interest.
Purity: Greater than 90%, as determined by SDS-PAGE.
Aggregation: Less than 10%, as determined by HPLC.
Filtration: 0.2 µm post-manufacturing filtered.
靶标信息
The CD8B antigen is a cell surface glycoprotein found on most cytotoxic T lymphocytes that mediates efficient cell-cell interactions within the immune system. The CD8 antigen, acting as a coreceptor, and the T-cell receptor on the T lymphocyte recognize antigens displayed by an antigen presenting cell (APC) in the context of class I MHC molecules. The functional coreceptor is either a homodimer composed of two alpha chains, or a heterodimer composed of one alpha and one beta chain. Both alpha and beta chains share significant homology to immunoglobulin variable light chains. This gene encodes the CD8 beta chain isoforms. Multiple alternatively spliced transcript variants encoding distinct membrane associated or secreted isoforms have been described. A pseudogene, also located on chromosome 2, has been identified.
仅用于科研。不用于诊断过程。未经明确授权不得转售。
生物信息学
蛋白别名: CD8; CD8a; CD8alpha; CD8b; CD8beta; Leu-2; Lymphocyte antigen 3; MAL; p32; T-cell membrane glycoprotein Ly-3; T-cell surface glycoprotein CD8 beta chain; T-cell surface glycoprotein Lyt-3
基因别名: Cd8b; Cd8b1; Ly-3; Ly-C; Lyt-3; Lyt3
UniProt ID: (Mouse) P10300
Entrez Gene ID: (Mouse) 12526