Search
Thermo Scientific Chemicals
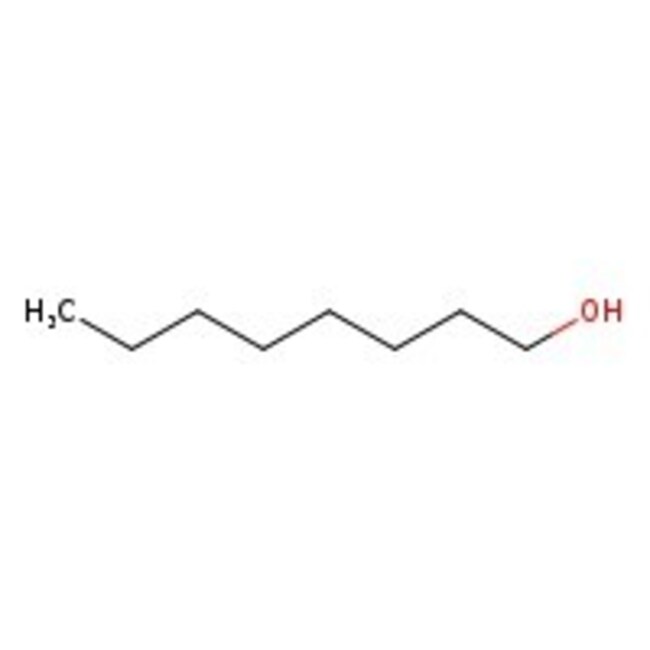
1-Octanol, natural, 98%
CAS: 111-87-5 | C8H18O | 130.23 g/mol
化学标识符
CAS111-87-5
IUPAC Nameoctan-1-ol
Molecular FormulaC8H18O
InChI KeyKBPLFHHGFOOTCA-UHFFFAOYSA-N
SMILESCCCCCCCCO
查看更多内容
规格 规格表
规格表
Identification (FTIR)Conforms
FormLiquid
Assay (GC)≥97.5%
Refractive Index1.4270-1.4310 @ 20?C
Appearance (Color)Clear colorless
1-Octanol is a synthetic reagent used to produce esters. It inhibits T-type calcium channels and is used to determine the partition coefficient between compounds and the cytosol. It is used to evaluate the lipophilicity of pharmaceutical products. It is also used as a solvent for protective coatings, waxes, and oils, and as a raw material for plasticizers.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
1-Octanol is a synthetic reagent used to produce esters. It inhibits T-type calcium channels and is used to determine the partition coefficient between compounds and the cytosol. It is used to evaluate the lipophilicity of pharmaceutical products. It is also used as a solvent for protective coatings, waxes, and oils, and as a raw material for plasticizers.
Solubility
Not miscible in water.
Notes
Stable under recommended storage conditions. Incompatible with oxidizing agents, acids, acid chlorides and acid anhydrides.
1-Octanol is a synthetic reagent used to produce esters. It inhibits T-type calcium channels and is used to determine the partition coefficient between compounds and the cytosol. It is used to evaluate the lipophilicity of pharmaceutical products. It is also used as a solvent for protective coatings, waxes, and oils, and as a raw material for plasticizers.
Solubility
Not miscible in water.
Notes
Stable under recommended storage conditions. Incompatible with oxidizing agents, acids, acid chlorides and acid anhydrides.
RUO – Research Use Only
General References:
- Hiromi Yamashita; Yuichi Ichihashi; Masaru Harada; Gina Stewart; Marye Anne Fox; Masakazu Anpo. Photocatalytic degradation of 1-octanol on anchored titanium oxide and on TiO2 powder catalysts. Journal of Catalysis. 1996, 158,(1), 97-101.
- Tai-Horng Young; Liao-Ping Cheng; Dar-Jong Lin; Ling Fane; Wen-Yuan Chuang. Mechanisms of PVDF membrane formation by immersion-precipitation in soft (1-octanol) and harsh (water) nonsolvents. Polymer. 1999, 40,(19), 5315-5323.