Search
Thermo Scientific Chemicals
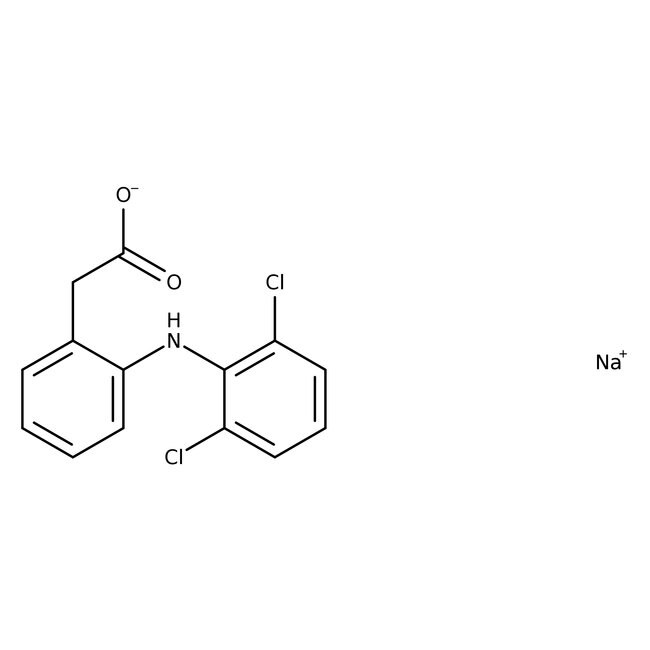
Diclofenac sodium salt
Standard NSAID and cyclooxygenase (COX) inhibitor | CAS: 15307-79-6 | C14H10Cl2NNaO2 | 318.129 g/mol
化学标识符
CAS15307-79-6
Oxidation of diclofenac sodium produces the metabolite 4'-hydroxy diclofenac) which demonstrates specific inhibition of Cox-2. Inhibition of Cox by diclofenac and 4'-hydroxy diclofenac suppresses prostaglandin E2 synthesis, producing anti-inflammatory and analgesic effects. Diclofenac is also shown to stabilize the native tetrameric conformation of transthyretin (TTR) fibrils, preventing the formation of insoluble amyloidogenic TTR deposits. Diclofenac Sodium is a substrate of CYP2C9. It is also used as an inhibitor of Cox-1 and Cox-2.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Oxidation of diclofenac sodium produces the metabolite 4′-hydroxy diclofenac) which demonstrates specific inhibition of Cox-2. Inhibition of Cox by diclofenac and 4′-hydroxy diclofenac suppresses prostaglandin E2 synthesis, producing anti-inflammatory and analgesic effects. Diclofenac is also shown to stabilize the native tetrameric conformation of transthyretin (TTR) fibrils, preventing the formation of insoluble amyloidogenic TTR deposits. Diclofenac Sodium is a substrate of CYP2C9. It is also used as an inhibitor of Cox-1 and Cox-2.
Solubility
Soluble in water (50 mg/ml), PBS pH 7.2 (6 mg/ml), ethanol (∼35 mg/ml), DMF (∼35 mg/ml), DMSO (∼35 mg/ml), methanol (>24 mg/ml), and acetone (6 mg/ml).
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Keep away from strong oxidizing agents.
Oxidation of diclofenac sodium produces the metabolite 4′-hydroxy diclofenac) which demonstrates specific inhibition of Cox-2. Inhibition of Cox by diclofenac and 4′-hydroxy diclofenac suppresses prostaglandin E2 synthesis, producing anti-inflammatory and analgesic effects. Diclofenac is also shown to stabilize the native tetrameric conformation of transthyretin (TTR) fibrils, preventing the formation of insoluble amyloidogenic TTR deposits. Diclofenac Sodium is a substrate of CYP2C9. It is also used as an inhibitor of Cox-1 and Cox-2.
Solubility
Soluble in water (50 mg/ml), PBS pH 7.2 (6 mg/ml), ethanol (∼35 mg/ml), DMF (∼35 mg/ml), DMSO (∼35 mg/ml), methanol (>24 mg/ml), and acetone (6 mg/ml).
Notes
Store in cool place. Keep container tightly closed in a dry and well-ventilated place. Keep away from strong oxidizing agents.
RUO – Research Use Only
General References:
- Barnett, J., et al. Purification, characterization and selective inhibition of human prostaglandin G/H synthase 1 and 2 expressed in the baculovirus system. Biochim. Biophys. Acta. 1994, 1209, (1), 130-9.
- Laneuville, O., et al. Differential inhibition of human prostaglandin endoperoxide H synthases-1 and -2 by nonsteroidal anti-inflammatory drugs. J. Pharmacol. Exp. Ther. 1994, 271, (2), 927-34.