Search
Thermo Scientific Chemicals
乙酸异丙酯, 99+%
CAS: 108-21-4 | C5H10O2 | 102.133 g/mol
| 货号 | 数量 |
|---|---|
| L09337.0F 又称 L09337-0F | 2500 mL |
化学标识符
CAS108-21-4
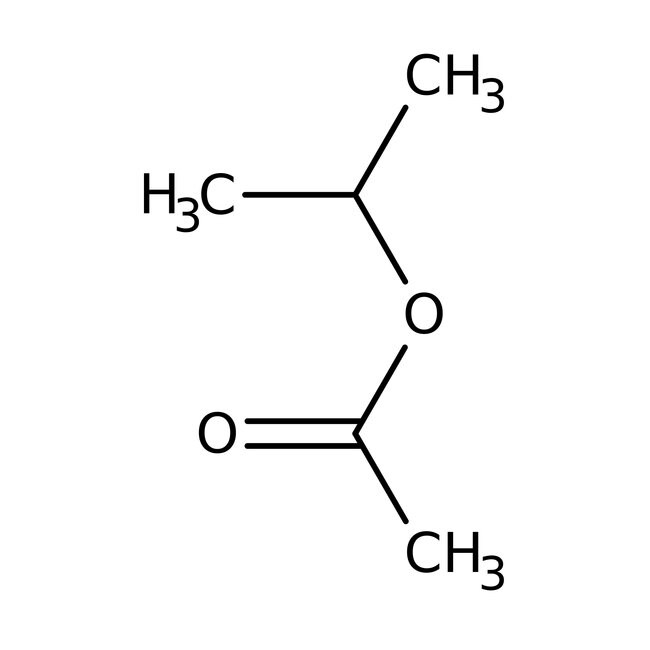
IUPAC Namepropan-2-yl acetate
Molecular FormulaC5H10O2
InChI KeyJMMWKPVZQRWMSS-UHFFFAOYSA-N
SMILESCC(C)OC(C)=O
查看更多内容
规格 规格表
规格表
Assay (GC)≥99.0% (UK-sourced material)
Identification (FTIR)Conforms (UK-sourced material)
Refractive Index1.3755-1.3785 @ 20?C (UK-sourced material)
Appearance (Color)Clear colorless
Transition Temperature≥99.0% (US-sourced material)
查看更多内容
Isopropyl acetate is a solvent in chemical industry, especially for cellulose, plastics, waxes, resins, gums, paints, oil and fats. and also as flavoring agent. It is an active component of perfumes and printing inks. It is also employed as an extractant for the preparation of antibiotics, vitamins and hormones.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Isopropyl acetate is a solvent in chemical industry, especially for cellulose, plastics, waxes, resins, gums, paints, oil and fats. and also as flavoring agent. It is an active component of perfumes and printing inks. It is also employed as an extractant for the preparation of antibiotics, vitamins and hormones.
Solubility
Slightly miscible with cold water and methanol.
Notes
Moisture sensitive. Incompatible with strong oxidizing agents, strong acids and bases.
Isopropyl acetate is a solvent in chemical industry, especially for cellulose, plastics, waxes, resins, gums, paints, oil and fats. and also as flavoring agent. It is an active component of perfumes and printing inks. It is also employed as an extractant for the preparation of antibiotics, vitamins and hormones.
Solubility
Slightly miscible with cold water and methanol.
Notes
Moisture sensitive. Incompatible with strong oxidizing agents, strong acids and bases.
RUO – Research Use Only
General References:
- Suggested replacement for chlorinated solvents in some applications, e.g. in peptide coupling reactions: J. Org. Chem., 60, 3569 (1995).
- Qiu, T.; Zhang, P.; Yang, J.; Xiao, L.; Ye, C. Novel Procedure for Production of Isopropanol by Transesterification of Isopropyl Acetate with Reactive Distillation. Ind. Eng. Chem. Res. 2014, 53 (36), 13881-13891.
- Akyalcın, S. Kinetic Study of the Formation of Isopropyl Alcohol by Transesterification of Isopropyl Acetate with Methanol in the Presence of Heterogeneous Catalyst. Ind. Eng. Chem. Res. 2014, 53 (23), 9631-9637.