Search
Thermo Scientific Chemicals
六氟磷酸,60% w/w 水溶液
CAS: 16940-81-1 | HPF6
| 货号 | 数量 |
|---|---|
| L15728.AE 又称 L15728-AE | 100 mL |
化学标识符
CAS16940-81-1
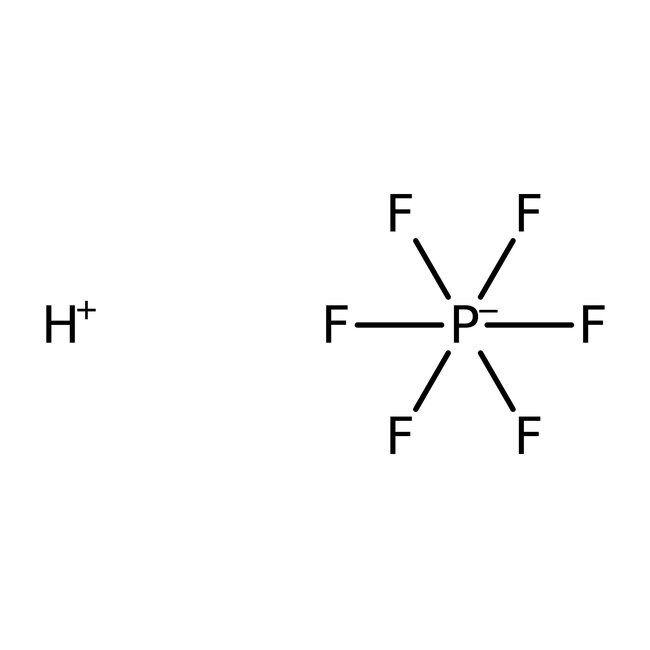
IUPAC Namehydrogen hexafluoro-λ⁵-phosphanuide
Molecular FormulaF6HP
InChI KeyLJQLCJWAZJINEB-UHFFFAOYSA-O
SMILES[H+].F[P-](F)(F)(F)(F)F
查看更多内容
规格 规格表
规格表
Assay (unspecified)55-65%
Appearance (Color)Clear, colorless to pale pink or pale yellow
FormLiquid
Hexafluorophosphoric acid is used as a catalyst. It is also used as a metal cleaner and chemical polishing agent for the formation of protective coatings of metal surfaces..
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Hexafluorophosphoric acid is used as a catalyst. It is also used as a metal cleaner and chemical polishing agent for the formation of protective coatings of metal surfaces..
Solubility
Immiscible with water.
Notes
Store in a cool place. Incompatible with strong bases, glass, alkali metals, powdered metals, strong oxidizing agents, metals and reacts violently with water.
Hexafluorophosphoric acid is used as a catalyst. It is also used as a metal cleaner and chemical polishing agent for the formation of protective coatings of metal surfaces..
Solubility
Immiscible with water.
Notes
Store in a cool place. Incompatible with strong bases, glass, alkali metals, powdered metals, strong oxidizing agents, metals and reacts violently with water.
RUO – Research Use Only
General References:
- Thermal decomposition of diazonium hexafluorophosphates often gives better results in the synthesis of aryl fluorides than the classical Balz-Schiemann method via the tetrafluoroborates (see Tetrafluoroboric acid, L14037). For an example, see: Org. Synth. Coll., 5, 133 (1973).
- Bedouret, L.; Judeinstein, P.; Ollivier, J.; Combet, J.; Desmedt, A. Proton Diffusion in the Hexafluorophosphoric Acid Clathrate Hydrate. J. Phys. Chem. B 2014, 118 (47), 13357-13364.
- Maiti, J.; Kakati, N.; Lee, S. H.; Yoon, Y. S. Fluorination of multiwall carbon nanotubes by a mild fluorinating reagent HPF 6. J. Fluorine Chem. 2012, 135, 362-366.