Search
Thermo Scientific Chemicals
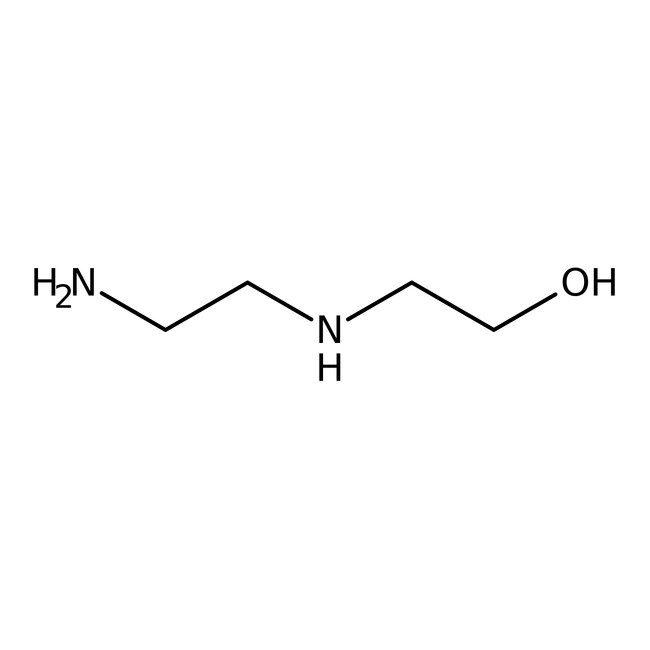
N-(2-羟乙基)乙二胺, 99%
CAS: 111-41-1 | C4H12N2O | 104.15 g/mol
化学标识符
CAS111-41-1
IUPAC Name2-[(2-aminoethyl)amino]ethan-1-ol
Molecular FormulaC4H12N2O
InChI KeyLHIJANUOQQMGNT-UHFFFAOYSA-N
SMILESNCCNCCO
查看更多内容
规格 规格表
规格表
Appearance (Color)Clear colorless to yellow
FormLiquid
Assay (Non-aqueous acid-base Titration)≥98.5 to ≤101.5% (non-U.S. specification)
Assay≥98.5% (U.S. specification)
CommentSpecification differs for U.S. and non-U.S. material where indicated
查看更多内容
It is used as an important intermediate in the manufacture of lube oil additives, fuel additives, chelating agents, surfactants and fabric softeners among other applications. N-(2-Hydroxyethyl)ethylenediamine [N-(2-Aminoethyl)ethanolamine] is used to study the aerobic biodecomposition of amines in hypersaline wastewaters.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
It is used as an important intermediate in the manufacture of lube oil additives, fuel additives, chelating agents, surfactants and fabric softeners among other applications. N-(2-Hydroxyethyl)ethylenediamine [N-(2-Aminoethyl)ethanolamine] is used to study the aerobic biodecomposition of amines in hypersaline wastewaters.
Solubility
Fully miscible in water.
Notes
Air Sensitive & Hygroscopic. Store under inert gas. Keep away from incompatibles.
It is used as an important intermediate in the manufacture of lube oil additives, fuel additives, chelating agents, surfactants and fabric softeners among other applications. N-(2-Hydroxyethyl)ethylenediamine [N-(2-Aminoethyl)ethanolamine] is used to study the aerobic biodecomposition of amines in hypersaline wastewaters.
Solubility
Fully miscible in water.
Notes
Air Sensitive & Hygroscopic. Store under inert gas. Keep away from incompatibles.
RUO – Research Use Only
General References:
- Hassan Ali Zamani.; Mohammad Taghi Hamed-Mosavian.;Elham Hamidfar.; Mohammad Reza Ganjali.; Parviz Norouzi. A novel iron (III)-PVC membrane potentiomeric sensor based on N-(2-hydroxyethyl)ethylenediamine-N,N',N″-triacetic acid. Materials Science and Engineering: C. 2008, 28, (8), 1551-1555.
- Hiroshi Ogino.; Mamoru Shimoi.; Yoko Saito. Structural identification of the reactive vanadium(III) intermediate formed in the electron-transfer reactions of [N'-(2-hydroxyethyl)ethylenediamine-N,N,N'-triacetato]aquavanadium(III) complex ([V(hedtra)(H2O)]) with halopentaamminecobalt(III) complexes: x-ray crystal structures of [V(hedtra)(H2O)].cntdot.2H2O and K[VO(hedtra)].cntdot.H2O. norg. Chem 1989, 28, (18), 3596-3600.