Search
Thermo Scientific Chemicals
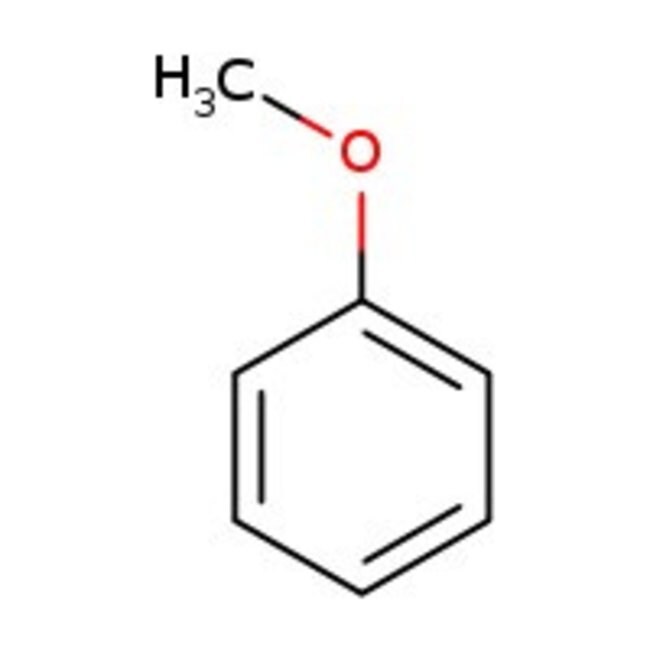
苯甲醚, 99%
CAS: 100-66-3 | C7H8O | 108.14 g/mol
| 货号 | 数量 |
|---|---|
| A12997.36 又称 A12997-36 | 500 g |
化学标识符
CAS100-66-3
IUPAC Nameanisole
Molecular FormulaC7H8O
InChI KeyRDOXTESZEPMUJZ-UHFFFAOYSA-N
SMILESCOC1=CC=CC=C1
查看更多内容
规格 规格表
规格表
Assay from Supplier's CofA≥98.5% (US Sourced Material)
Refractive Index1.5155-1.5185 @ 20?C
CommentMaterial Sourced in UK and US
Identification (FTIR)Conforms (UK Sourced Material)
Appearance (Color)Clear colorless
查看更多内容
Anisole is widely used as a solvent for the synthesis of various organic compounds, anethole, nonylphenol isomer 4-(3',6'-dimethyl-3-heptyl)phenol, perfumes, insect pheromones and pharmaceuticals. It finds application in the preparation of inorganic complexes and materials such as tin-core/tin oxide nanoparticles.
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo Scientific Chemicals.
Applications
Anisole is widely used as a solvent for the synthesis of various organic compounds, anethole, nonylphenol isomer 4-(3′,6′-dimethyl-3-heptyl)phenol, perfumes, insect pheromones and pharmaceuticals. It finds application in the preparation of inorganic complexes and materials such as tin-core/tin oxide nanoparticles.
Solubility
Miscible with methanol and acetone. Immiscible with water.
Notes
Keep away from sources of ignition. Incompatible with strong oxidizing agents.
Anisole is widely used as a solvent for the synthesis of various organic compounds, anethole, nonylphenol isomer 4-(3′,6′-dimethyl-3-heptyl)phenol, perfumes, insect pheromones and pharmaceuticals. It finds application in the preparation of inorganic complexes and materials such as tin-core/tin oxide nanoparticles.
Solubility
Miscible with methanol and acetone. Immiscible with water.
Notes
Keep away from sources of ignition. Incompatible with strong oxidizing agents.
RUO – Research Use Only
General References:
- The rate of ortho-lithiation of anisole is accelerated dramatically in the presence of TMEDA, effective also in sub-stoichiometric amounts: Tetrahedron Lett., 35, 385 (1994). For discussion of the mechanism of ortho-lithiation, see: J. Org. Chem., 62, 3024 (1997). Although direct dilithiation with n-BuLi/TMEDA was unsuccessful, a sequence involving monolithiation, silylation and further lithiation at the 6-position gave the 2,6-disilyl derivative in 72% yield: J. Org. Chem., 49, 4657 (1984).
- Widely used as a carbocation scavenger in the deblocking of, e.g. N-Boc or N-Cbz (Z) peptides with acidic reagents. Addition of anisole removes t-butyl or benzyl cations from the reaction mixture; see, e.g.: J. Chem. Soc., Chem. Commun., 922 (1976); Chem. Pharm. Bull., 25, 2929 (1977); Chem. Ber., 101, 3649 (1968).
- Lee, W-S.; Kumar, A.; Wang, Z.; Bhan, A. Chemical Titration and Transient Kinetic Studies of Site Requirements in Mo2C-Catalyzed Vapor Phase Anisole Hydrodeoxygenation. ACS Cata. 2015, 5 (7), 4104-4114.
- Peters, J. E.; Carpenter, J. R.; David, D. C. Anisole and Guaiacol Hydrodeoxygenation Reaction Pathways over Selected Catalysts. Energy Fuels 2015, 29 (2), 906-916.