Search
引用和文献 (13)
Invitrogen™
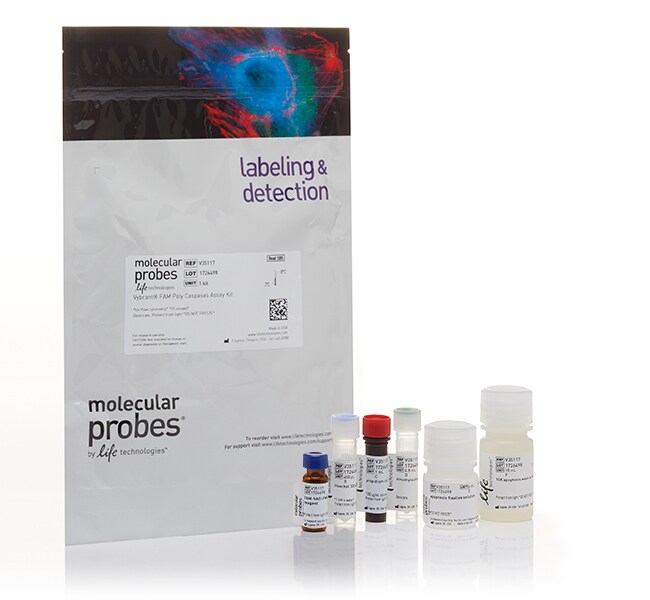
用于流式细胞分析的 Vybrant™ FLICA 半胱天冬酶细胞凋亡检测试剂盒
采用用于检测多聚半胱天冬酶和半胱天冬酶-3/7/8 活性的 Vybrant FLICA 半胱天冬酶检测试剂盒,通过流式细胞分析检测细胞凋亡情况。
| 货号 | 酶 |
|---|---|
| V35119 | Caspase 8 |
| V35117 | Poly Caspases |
| V35118 | Caspase 3/7 |
货号 V35119
价格(CNY)
3,516.00
飞享价
Ends: 31-Dec-2026
4,671.00共减 1,155.00 (25%)
25 assays
酶:
Caspase 8
价格(CNY)
3,516.00
飞享价
Ends: 31-Dec-2026
4,671.00共减 1,155.00 (25%)
25 assays
使用适用于多聚半胱天冬酶以及半胱天冬酶 3 和 7 的 Vybrant FAM 半胱天冬酶检测试剂盒,通过流式细胞分析,快速检测半胱天冬酶介导的细胞凋亡。Vybrant FAM 半胱天冬酶检测试剂盒使用荧光半胱天冬酶抑制剂 (FLICA) 方法,用于检测和报告半胱天冬酶活性。具体而言,这些半胱天冬酶检测试剂盒包含经工程改造的 FLICA 底物 FAM-VAD-FMK(用于多聚半胱天冬酶)、FAM-DEVD-FMK(用于半胱天冬酶-3/7)和 FAM-LETD-FMK(用于半胱天冬酶-8)。它们还包括 Hoechst 33342 和碘化丙啶染色剂,能够通过流式细胞分析同时评估膜可透过性和细胞周期。
用于流式细胞分析的 Vybrant FAM 半胱天冬酶检测试剂盒采用了一种检测活性半胱天冬酶的新方法:该试剂盒利用 FLICA 方法(亲和标记)。该亲和标记将一个可以与半胱氨酸发生共价反应的氟甲基酮 (FMK) 部分和一个半胱天冬酶特异性氨基酸序列相连接。不同的氨基酸识别序列用于检测不同的半胱天冬酶:缬氨酸-丙氨酸-天冬氨酸 (VAD) 用于检测多聚半胱天冬酶(包括半胱天冬酶-1、-3、-4、-5、-6、-7、-8 和 -9),天冬氨酸-谷氨酸-缬氨酸-天冬氨酸 (DEVD) 用于检测半胱天冬酶-3/7,亮氨酸-谷氨酸-苏氨酸-天冬氨酸 (LETD) 用于检测半胱天冬酶-8。连有一个羧基荧光素 (FAM) 基团,作为报告基团。
FLICA 试剂与已激活半胱天冬酶的反应中心通过识别序列相互作用,之后通过 FMK 基团与半胱氨酸共价连接。FLICA 抑制剂具有细胞渗透性但没有细胞毒性。未结合的 FLICA 分子扩散出细胞并被洗涤掉,其余的荧光信号可用于直接测定加入抑制剂时存在的活性半胱天冬酶的量。FLICA、碘化丙啶和 Hoechst 33342 试剂的近似激发峰和发射峰分别为 488 nm/⁄530 nm、535⁄ nm/6617 nm 和 350 nm/⁄461 nm。Vybrant FAM 多聚半胱天冬酶检测、半胱天冬酶-3 和半胱天冬酶-7 检测以及半胱天冬酶-8 检测细胞凋亡试剂盒中包括的检测试剂分别为 488⁄⁄530 FAM-VAD-FMK 试剂、488/⁄530 FAM-DEVD-FMK 和 488/⁄530 FAM-LETD-FMK。
FLICA 试剂与已激活半胱天冬酶的反应中心通过识别序列相互作用,之后通过 FMK 基团与半胱氨酸共价连接。FLICA 抑制剂具有细胞渗透性但没有细胞毒性。未结合的 FLICA 分子扩散出细胞并被洗涤掉,其余的荧光信号可用于直接测定加入抑制剂时存在的活性半胱天冬酶的量。FLICA、碘化丙啶和 Hoechst 33342 试剂的近似激发峰和发射峰分别为 488 nm/⁄530 nm、535⁄ nm/6617 nm 和 350 nm/⁄461 nm。Vybrant FAM 多聚半胱天冬酶检测、半胱天冬酶-3 和半胱天冬酶-7 检测以及半胱天冬酶-8 检测细胞凋亡试剂盒中包括的检测试剂分别为 488⁄⁄530 FAM-VAD-FMK 试剂、488/⁄530 FAM-DEVD-FMK 和 488/⁄530 FAM-LETD-FMK。
仅供科研使用。不可用于诊断程序。
规格
酶Caspase 8
激发/发射FAM-LETD-FMDK:488/530 PI:535/617 Hoechst 33342:350/461
流式细胞仪激光线路UV,488
适用于(设备)流式细胞仪
标签类型其他标记或染料
标签或染料FAM, propidium iodide, Hoechst 33342
产品线Vybrant
产品类型半胱天冬酶检测试剂盒
数量25 Assays
运输条件室温
检测方法荧光
产品规格管装
Unit Size25 assays
内容与储存
1 小瓶 FAM-LETD-FMK 半胱天冬酶-8 试剂(FLICA 试剂)、1 小瓶 Hoechst 33342、1 小瓶碘化丙啶、1 小瓶 DMSO、1 瓶细胞凋亡固定剂溶液和 1 瓶细胞凋亡洗涤缓冲液(10X 溶液)。避光储存在冷藏冰箱 (2-8°C) 中。
引用和文献 (13)
引用和文献
Abstract
Galectin-3 binds to CD45 on diffuse large B-cell lymphoma cells to regulate susceptibility to cell death.
Journal:Blood
PubMed ID:23065155
Diffuse large B-cell lymphoma (DLBCL) is the most common non-Hodgkin lymphoma and an aggressive malignancy. Galectin-3 (gal-3), the only antiapoptotic member of the galectin family, is overexpressed in DLBCL. While gal-3 can localize to intracellular sites, gal-3 is secreted by DLBCL cells and binds back to the cell surface in
Reovirus infection or ectopic expression of outer capsid protein micro1 induces apoptosis independently of the cellular proapoptotic proteins Bax and Bak.
Journal:J Virol
PubMed ID:20980509
Mammalian orthoreoviruses induce apoptosis in vivo and in vitro; however, the specific mechanism by which apoptosis is induced is not fully understood. Recent studies have indicated that the reovirus outer capsid protein µ1 is the primary determinant of reovirus-induced apoptosis. Ectopically expressed µ1 induces apoptosis and localizes to intracellular membranes.
Interrelated roles for Mcl-1 and BIM in regulation of TRAIL-mediated mitochondrial apoptosis.
Journal:J Biol Chem
PubMed ID:16478725
The current study demonstrates a novel cross-talk mechanism between the TRAIL receptor death signaling pathway and the mitochondria. This newly identified pathway is regulated at the mitochondrial outer membrane by a complex between the prosurvival Bcl-2 member, Mcl-1 and the BH3-only protein, Bim. Under non-apoptotic conditions, Bim is sequestered by
Assay of caspase activation in situ combined with probing plasma membrane integrity to detect three distinct stages of apoptosis.
Journal:J Immunol Methods
PubMed ID:12072182
Activation of cysteine-aspartic acid specific proteases (caspases) in situ, in live cells, can be detected using fluorochrome-labeled inhibitors of caspases (FLICA), the reagents that covalently bind to the active center of these enzymes. In the present study, this assay was combined with a probe of plasma membrane capacity to exclude
Interactions of fluorochrome-labeled caspase inhibitors with apoptotic cells: a caution in data interpretation.
Journal:Cytometry A
PubMed ID:12938188
BACKGROUND: Fluorochrome-labeled inhibitors of caspases (FLICA, e.g., FAM-VAD-FMK, FITC-VAD-FMK) have been designed as affinity labels of the enzyme active center of caspases Their binding by apoptotic cells was interpreted as reflecting activation of caspases. We have recently observed, however, that their binding is more complex and may involve additional mechanisms.