Search
- Home
- Shop All Products
- Electrolytes for battery research
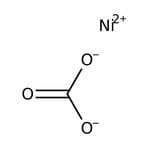
碳酸镍(II),无水, 98%
Nickel(II) carbonate is used in sulphamate baths, metal phosphating, electroplating and ceramic applications. It acts as a precursor to catalysts and an intermediate in the hydrometallurgical purification of nickel from its ores.

氧化钴(II,III) , 纳米粉, 99% (metals basis)
Cobalt(II,III) oxide is used in enamels, pigments, porcelain colorants, semiconductors, ceramics, catalysis, grinding wheels and other fields as an important inorganic material. It is a useful oxidant in chemical industry and as catalyst for organic synthesis.

氧化镍(II), 99% (metals basis)
It is used in nickel-iron battery (Edison Battery) and fuel cells. It is a precursor for many nickel salts and nickel alloys including nickel-steel allys, for example invar and elinvar. It is used in electrochromic devices, frits, ferrites, and porcelain glazes.
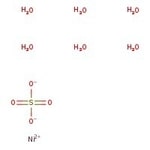
硫酸镍(II)六水合物, 98%
Nickel sulfate hexahydrate is mainly used in Ni plating, as mordant in dyeing and in printing textiles, as a blackening agent for zinc and brass and in the fabrication of organic nickel salts. It is used as an effective catalyst for gas-phase dehydration of glycerol to acrolein.
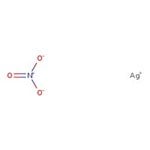
硝酸银, ACS, 99.9+% (metals basis)
Silver nitrate is employed in Silver plating, photography, manufacturing of other silver compounds, mirrors, coloring porcelain, and staining in histology. It finds use as a stain in scanning electron microscopy, and a key ingredient in several silver-based antiseptic and antibacterial...
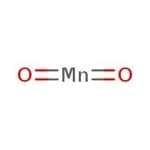
二氧化锰, Puratronic®, 99.996% (metals basis)
It is used as an oxidizing agent in organic synthesis such as oxidation of allylic/benzylic alcohols, as a textile dye, as a reducing agent, and as a component of dry cell batteries such as zinc-carbon battery and alkaline battery.

三氯化铁(III) 无水, 98%
1-methyl-2-pyrrolidinone, is useful for spectrophotometry, chromatography and ICP-MS detection. It is useful for the chemical functionalization of graphene sheets by solvothermal reduction of graphene oxide.

二氧化锰(IV), 99.9% (metals basis)
It is used as an oxidizing agent in organic synthesis such as oxidation of allylic/benzylic alcohols, as a textile dye, as a reducing agent, and as a component of dry cell batteries such as zinc-carbon battery and alkaline battery.
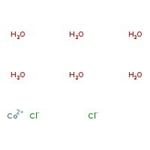
六水合氯化钴(II), 99.9% (metals basis)
Cobalt(II) chloride hexahydrate is used as a humidity indicator in weather instruments. It finds use in electroplating of cobalt, in invisible ink, in organic chemistry and is a precursor to cobaltocene(bis (cyclopentadienyl) cobalt(II)).
无水氯化铜(II), 98%最低
Glassy carbon is widely practiced as an electrode material in electrochemistry, as considerably as for high temperature crucibles and as a part of some prosthetic devices. This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio.
六水合硝酸钴(II), ACS, 98.0-102.0%
This Thermo Scientific Chemicals brand product was originally part of the Alfa Aesar product portfolio. Some documentation and label information may refer to the legacy brand. The original Alfa Aesar product / item code or SKU reference has not changed as a part of the brand transition to Thermo...
六水合硝酸钇(III), 99.9% (REO)
It is used as a reagent, in optics, ceramics, glass and electronics. It is used in superconducting materials. It has been reported as a reusable catalyst in Bignelli reaction for the synthesis of heterocycles such as pyrimidin-2-ones.