Search

产品简介
电雾式检测具有高灵敏度,并且提供近乎通用的检测,独立于分析物结构。由于所有行业中具有这些独特的特性,液相色谱 仪在低波长 UV ,蒸发光散射检测器 (ELSD) 和折射率 (RID) 等其他通用检测器上经常使用电雾式检测器 (CAD)。
与每个 LC 检测器一样,您应该了解和解决一些关键问题,以优化 CAD 的性能。对 CAD 的工作原理以及哪些因素会对性能产生负面影响有基本了解,这对于生成高质量,可靠的数据以及防止检测器损坏至关重要。
在优化电雾式检测器的性能时,要考虑多个方面,更重要的是保存均匀的分析物响应。分析物挥发性,流动相质量和成分以及盐形成等物质对一致响应的影响最大。
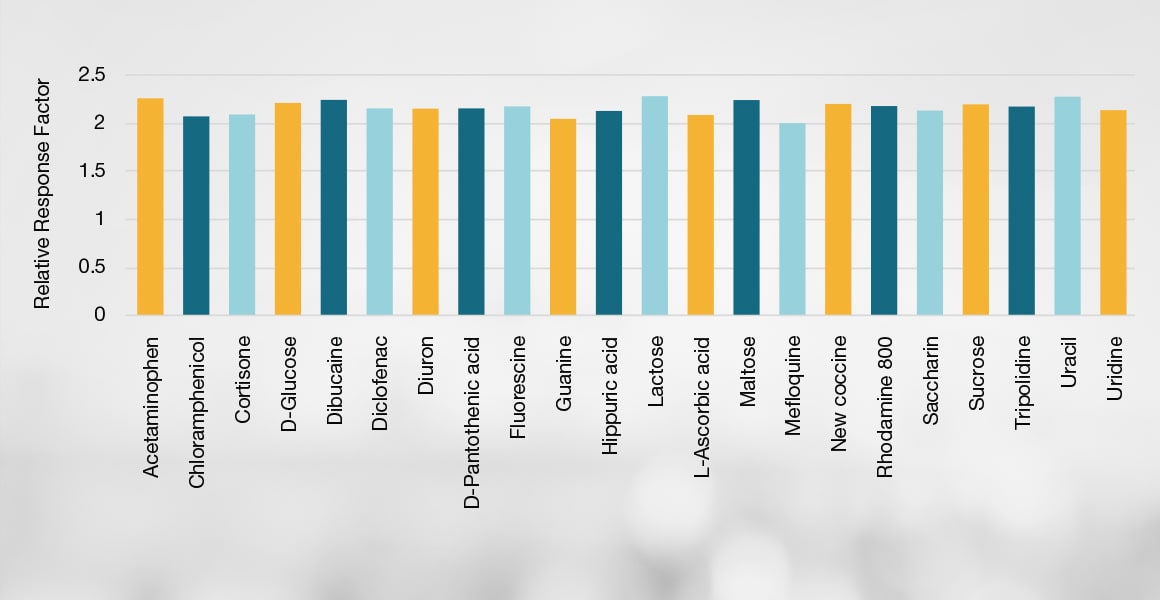
影响 CAD 响应的因素汇总
| 系数 | 考虑因素 | 对 CAD 响应的影响 |
| 淋洗液流 | 在流动相中引入半挥发性或非挥发性成分 | 增加背景信号,噪声和漂移。非挥发性添加剂可能会严重损坏 CAD |
| 使用具有最低蒸发残留的优质添加剂 | 建议使用 LCMS 级添加剂来降低基线噪声 | |
| 离子分析物和带电添加剂之间的盐形成 | 去除均一的分析物响应,可能会对检测器造成严重损坏 | |
| 通过梯度洗脱改变溶剂成分 | 可产生可重现的漂移。去除均一的分析物响应。溶剂成分的突然变化,例如步进梯度,会产生伪峰 | |
| 流动相质量和纯度 | 优选LCMS级溶剂。老化的溶剂会增加背景和噪音 | |
| 雾化 | 分析物挥发性 | 影响一致的分析物响应 |
| 气体流速与稳定性 | 氮气流速的任何变化都会增加基线信号。出于安全原因,氮气优于压缩空气,并且基线噪声降低 | |
| 气溶胶稳定性和效率 | 雾化器吸头处累积的盐可能影响液滴的形成 | |
| 蒸发温度 (EvapT) | 溶剂与分析物的干燥效率 | 增加 EvapT 可以改善背景信号和响应。降低 EvapT 可增加半挥发性物质的检测范围 |
| 其他因素 | 来自其他 LC 系统组件的非挥发性物质污染 | 增加背景,噪音和漂移。去除均一的分析物响应 |
| 实验室玻璃器皿中的残留肥皂和清洁剂 | 产生基线伪影和幽灵峰 | |
| 使用 pH 电极调节 pH | 给出基线伪影和幽灵峰 | |
| 浸出样品瓶和瓶盖中的化学品 | 生成基线伪影和幽灵峰 | |
| 标准质量和纯度 | 对纯度测量产生不利影响 | |
| 柱流失 / 分解 | 增加噪声和背景电流 |
分析物挥发性
由于给定分析物的 CAD 响应主要受气溶胶蒸发过程中化合物的挥发性影响,因此预测化合物是否是电雾式检测的良好候选物质的能力始于挥发性。这是开发 HPLC-CAD 方法的主要考虑因素。
与 CAD 兼容的化合物应至少显示以下物理特性之一:
- 沸点 > 400 °C
- 分子量 > 350 g/mol 和汽化 Enthpy > 64 kJ/mol
分析物在 CAD 兼容性方面的不稳定性一般规则是:
- 非挥发性分析物高度兼容,并提供类似均匀的响应。
- 半挥发性分析物往往表现出不均匀的响应,这取决于分析物的挥发性, EvapT 以及使用流动相添加剂形成盐的能力。
- 挥发性分析物显示无响应。
流动相组成
与 CAD 配合使用的任何流动相的一项重要前提是溶剂必须是易挥发性的,且不含任何非挥发性成分。
与流动相成分相关的几个因素会影响性能 ,更重要的是,响应一致性包括:
- 流动相质量和年龄
- 流动相添加剂和杂质
- 玻璃器皿和 pH 值调节
- 梯度洗脱
我们详细讨论这些因素,对 CAD 响应的影响以及准备流动阶段的最佳实践。
流动相制备
CAD 和 MS 的溶剂纯度要求非常相似。当您使用 质量差的流动相时, CAD 可能会显示高噪声,响应差,基线漂移和与梯度相关的伪迹峰。
为确保您的流动相清新且纯净,您可以遵循的一些最佳实践包括:
- 只能使用标有 LC/MS 级或更好的含有最低“蒸发后残留物”规格的溶剂。
- 对于所有商业溶剂,请记录批号和溶剂质量。记录蒸发后的残留物,以用于给定批次的溶剂。使用永久记号笔,记录接收和打开的日期,以避免无意中使用受污染或旧的溶剂。
- 如果使用的溶剂有不同的等级,供应商,批次等,请考虑使用质量已知的溶剂测试溶剂。
- 请勿将流动相储存以备日后使用,因为杂质和污染物会随时间推移而增加。
CAD 反应中产生高背景电流的常见原因可能来自用于流动相制备的水中的杂质。
- MΩ ASTM D1193 定义的 1 型超纯水,可在 25 °C 下使用主要规格 18.2 M Ω -cm 或更高的电阻率,上限使用 50 ppb 总有机碳, 1 ppb 钠, 1 ppb 氯化物和 3 ppb 总二氧化硅。
- 如果您无法获得 1 级超纯水,则我们建议使用建议使用含低杂质的优质瓶装水,如 UHPLC-MS 级水。请记住,使用瓶装水通常比新鲜超纯水产生更高的背景电流和噪声水平。
- 另一种方法是不使用纯水,而是使用低比例有机溶剂 (如甲醇或乙腈) 的水,但请确保根据这一变化调整梯度曲线。
大多数有机 HPLC 溶剂的沸点低于水,因此大多数溶剂满足流动相挥发性要求。但是许多溶剂等级包含大量的非挥发性杂质,这些杂质会对 CAD 性能产生不利影响,应避免使用。
- 一些等级的有机溶剂 (例如 THF) 通常用丁基羟基甲苯等添加剂进行稳定。
- 其他“高纯度”溶剂等级通常是为特定用途 (电化学或分光光度等级) 定制的,并经过优化,可包含尽可能少的特定类别杂质 (氧化还原活性或发色团污染物) ,但可能含有其他类别的大量杂质。
- 对于给定等级的溶剂,并非所有供应商都具有相同的“蒸发后残留物”规格。选择具有最低“蒸发后残留物”规格的溶剂等级有助于缓解所有这些问题,并使 CAD 保持最佳性能。
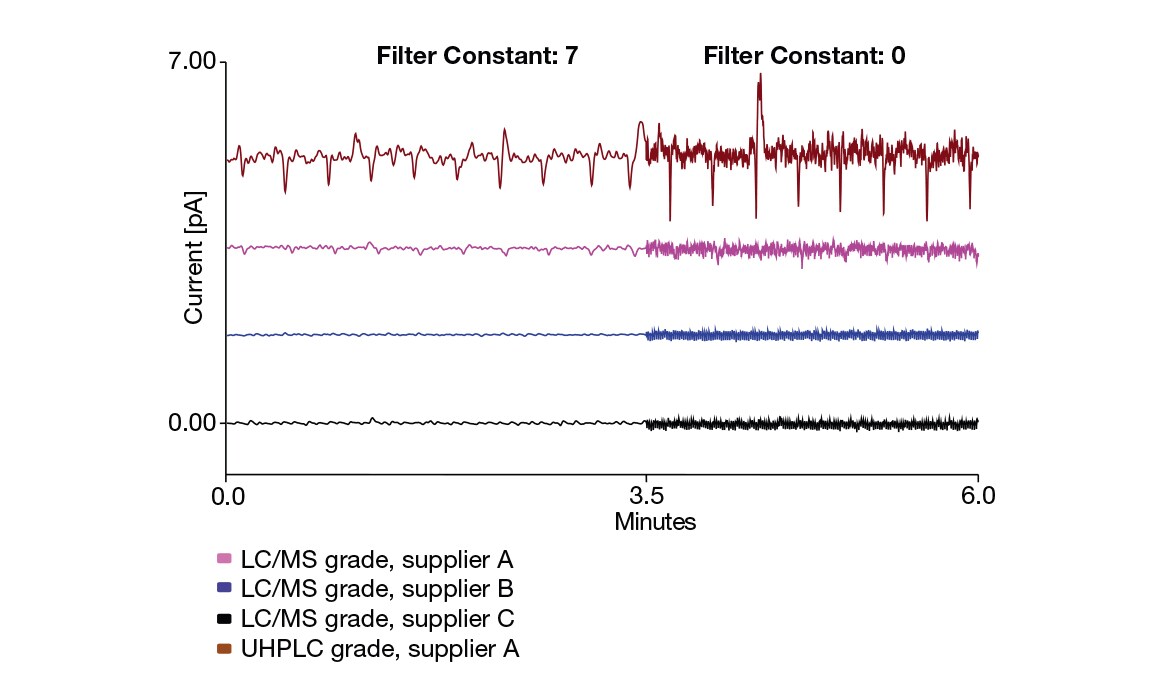
四种不同商业甲醇来源的 CAD 基线行为。黑色,蓝色和粉色痕迹– LC/MS 级;棕色– UHPLC 级。显示两个滤波常数:7 (0 – 3.5 分钟) 和 0 (3.5 – 6.0 分钟)。有关详细信息 ,请参阅技术说明 140 :优化和监测UV-VIS吸收、荧光和带电气溶胶探测器的溶剂质量。
您的实验室玻璃器皿是非挥发性污染物的常见来源,包括随时间推移从玻璃中浸出的化学品,先前使用的遗留残留物或清洁剂中的化学品。
- 仅使用专用于 CAD 的玻璃器皿,包括流动相储液槽,烧杯,烧瓶和刻度量筒。
- CAD 是分析去污剂和表面活性剂的绝佳工具,因此我们推荐无去污剂的清洁剂,手动清洗 CAD 专用玻璃器皿或在清洗后或使用前特别注意冲洗玻璃器皿。
- 始终使用合适的溶剂冲洗您的玻璃器皿,然后用高纯度去离子水三重冲洗,然后在流动相溶剂含有溶剂混合物时使用流动相溶剂冲洗或较弱洗脱流动相溶剂冲洗。
- 在清洁后让玻璃器皿风干 (避免使用纸巾) ,并覆盖任何未使用的开放式玻璃器皿以防止灰尘进入。
当调整流动相溶液的 pH 值时,请记住 pH 电极储存在氯化钾浓缩溶液中。电极表面残留液滴可能会给流动相带来可测量量的非挥发性盐,并可能通过提高背景噪声对 CAD 性能产生不利影响。
- 使用超纯水彻底冲洗电极表面。
- 避免将 pH 电极浸入流动相,使用小等份试液测量 pH 值并在 pH 调节完成后丢弃。
流动相添加剂
流动相添加剂通常包括缓冲液, pH 修饰剂和离子对试剂。如果您的 HPLC-CAD 方法需要流动相添加剂,则您需要考虑:
- 流动相中的半挥发性和非挥发性添加剂的浓缩。我们建议您将添加剂的浓度限制在达到所需分离效果的最低水平。
- 流动相添加剂中的杂质水平。更高水平的杂质通常会产生更高的背景电流,更高的基线噪声和更明显的基线漂移。
- 一些添加剂具有吸湿性或与空气发生反应。您的最佳选择是购买单个样品瓶使用的容器,并且仅使用新开封的样品瓶。
- 添加剂在雾化过程中与离子分析物形成盐的能力。这是在 (i) 尝试在添加剂和感兴趣离子之间形成盐以增强对半挥发性的响应; (ii) 避免在相位添加剂和带电污染物之间形成盐,从而导致较大的噪声和性能降低。
化学挥发性与 CAD 兼容性之间的关系
| 分析物 | 流动相 | 添加剂 | |
| 非易失性 | 高度兼容 | 不兼容 | 不兼容 |
| 半挥发性 | 兼容 | 兼容 | 兼容 |
| 易挥发的 | 不兼容 | 兼容 | 兼容 |
非挥发性流动相添加剂与 CAD 极其不兼容,并可能对性能产生不利影响。
- 短期暴露于非挥发性添加剂会提高噪声,降低响应和明显的基线漂移。
- 长期暴露,尤其是在高水平下,可能需要您的 CAD 进行重大服务或维修。
梯度洗脱
进入检测器的流动相的有机成分会影响雾化过程,并影响梯度洗脱过程中检测器响应的均匀性。
因此, CAD 响应取决于流动相组成,而流动成分的变化可能会影响统一响应和后续 定量能力。
通过将反 梯度工作流程应用在我们的反梯度 配置中,您可以克服梯度洗脱对 CAD 反应的影响的一种简单方法。该设置使用第二个泵产生第二个反向梯度,可恢复均一的分析物响应,并实现无标准定量。
成盐
通常,您会发现,使用流动相添加剂时背景电流和噪声会增加,这种效应会随着添加剂浓度的增加而增加,并随着添加剂挥发性的降低而降低。 如果您的方法需要添加剂,我们建议您使用最低级别的挥发性和半挥发性添加剂。但在某些情况下,盐的形成会增加 CAD 的反应。
以下是您应该了解的盐层结构:
1. 在某些情况下,不需要的盐形成会降低检测器性能
- 当盐从流动相中的离子化污染物中衍生而来
- 如果盐在流动相中形成的电离污染物和电离添加剂之间
- 当您将阴离子和阳离子配对试剂添加到流动相时
2. 有意盐的形成实际上会扩大 CAD 检测到的分析物范围
- 在离子对试剂和一种波荷电的半挥发性分析物之间形成盐,可迫使分析物表现出类似于非挥发性物质的行为。这在使用标准游离定量时很重要
挥发性添加剂和与 CAD 兼容的典型浓度
| 酸性添加剂 | mM | mg/mL | pKa | pH 值 |
| 乙酸 | 17.4 | 1.04 | 4.8 | 3.27 |
| 甲酸 | 26.3 | 1.09 | 3.8 | 2.7 |
| 三氟乙酸 | 13 | 1.48 | 0.0 | 1.9 |
| 缓冲液 | mM | mg/mL | pKa | 缓冲范围 |
乙酸铵 | 10 | 0.77 | 4.8 | 3.8-5.8. |
铵形式 | 10 | 0.63 | 3.8 | 2.8-4.8 |
*碳酸铵 | 10 | 0.96 | 10.3 9.3 7.8 | 7-11 |
* 基本流动相,特别是氨基流动相的问题是,随着流动阶段的空气逐渐形成碳酸盐。碳酸盐是非挥发性的,致检测器性能不佳。新鲜制作的流动相通常还好,但随着年龄的增长而退化。提高蒸发温度会显著减少问题,并允许在 CAD 中使用基本相。但是,确保色谱柱在使用基本相时保持稳定,否则色谱柱流失可能会有问题。
其他因素
特定色谱分析方法的标准品质量会影响结果的准确性,并影响 CAD 的噪声和背景电流。商业标准品的纯度水平通常由一种可能会遗漏某些杂质的影响的分析技术确定。
- 检查标签或参阅检验报告,以了解有关纯度测定所用分析方法的更多信息。
标准品可吸收大气中的水分 (吸湿性) 或吸收大气中的水分,然后溶解形成液体 (潮解性)。制备用于校准研究的标准品时,这两种问题都存在。其他化合物在暴露于大气中时可能不稳定,或在溶解于溶剂中时降解。要解决这些问题:
- 购买少量标准品,最好装在一次性密封样品瓶中,或使用预称重的密封安瓿瓶进行标准制备。
- 将标准品存储在活性干燥剂上,并在必要时更换。
- 在日志中记录您购买标准品的日期,打开时和每次使用后。
- 设置适当的到期日期,并在过期时更换。
当前版本的 CAD 与高达 2.0 mL/min 的流速和所有常用分析柱格式兼容。您应选择稳定的色谱柱,不含与方法温度和在水相条件附近的 pH 值要求兼容的流失。柱流失可引入半挥发性和非挥发性杂质,并可能提供更高的噪声和背景电流。
- 众所周知,某些色谱柱像硅胶,硅胶基质氨基和氰基;在可能时避免这些模式。
- 使用可提供良好分离度的最低色谱柱温度,有助于防止色谱柱流失。
样品瓶由两部分组成—主体和带隔垫的瓶盖—每一部分可能是产生基线伪峰和幽灵峰的污染来源。
在选择自动进样器样品瓶之前,您应检查材料类型是否对分析性能产生负面影响。用流动相填充样品瓶,并填充用于样品和标准品制备的各溶液。盖上,涡旋,停留约 72 小时。每天涡旋,确保试剂瓶盖暴露于溶液中。分析每个样品瓶的内容物并检查是否:
- 新的干扰峰出现在影响目标分析物峰的色谱图中。
- 溶液前部的变化会影响早期洗脱峰。
- 基线色谱图中的大扰动。
如果存在问题,您将需要测试其他样品瓶的兼容性。
如果没有问题,则表明您的样品瓶和瓶盖可以安全使用。
技术 CAD 资源
联系我们
*必填字段
你有问题吗?想了解更多关于带电气溶胶检测的信息吗?立即与解决方案专家交谈。









