Search
LC-MS Sample Preparation
Liquid chromatography mass spec sample preparation
Liquid chromatography mass spectrometry (LC/MS) is performed on sample types that are thermally unstable, large, polar, ionic or non-volatile, or which need to be derivatized. Typical LC/MS samples include nucleotides, peptides, steroids, hormones, dyes, fatty acids, and alcohols. As a result, LC/MS is preferentially used in pharmacokinetics, proteomics, metabolomics, lipidomics, and drug development.
Liquid chromatography is first used to separate sample components so that their relatively purified forms can be introduced into the mass spectrometer. Electrospray ionization (ESI) or atmospheric pressure chemical ionization (APCI) is typically applied to generate charged ions from the sample, and it is these ions that pass into the mass analyzer. The identity of the target ion is determined by comparing its mass:charge (m/z) ratio against a spectral database such as MassBank, METLIN and mzCloud.
LC/MS has some overlap with GC/MS, and LC/MS sample types can sometimes be analyzed by GC/MS, as the following figure shows:

Liquid chromatography mass spec sample preparation

LC/MS samples containing non-volatile or large compounds are typically prepared for mass spectrometry (MS) analysis by first undergoing liquid chromatography (LC) separation. This is a necessary step in the eventual isolation and identification of the target analyte from among hundreds, if not thousands, of other compounds and contaminants.
As opposed to the first gravity columns, today’s LC cartridges are packed with dense, fine-particle supports that permit analyte separation only via high pressure (HPLC) or ultra HPLC (UHPLC). Such advancement in chromatography technology has enabled the separation of molecules that differ from each other by only a few daltons. It has also challenged chromatographers to develop sample preparation methods that permit faster and cleaner liquid chromatography.
The sample’s physical and chemical characteristics are critical to determining how it is digested and prepared for liquid chromatography. Filtration is often applied to samples that contain particulates such as undissolved salts, and many laboratories use hydrophobic or hydrophilic PTFE filters to remove interfering materials and fine particles.
Physically, there is the analyte size to consider, which determines the pore and particle size of the cartridge used. Chemically, the pH of the analyte must be accounted for because this will determine the types of bonded phases (e.g., alkyl-bonded moiety) in the column’s packing material. Also, different columns are applicable to different sample pH ranges.
Total LC/MS sample preparation must take into consideration both the mobile phase (containing the analyte) and stationary phase (packing material or support). Soluble and insoluble matrix components should also be considered and whether they might interfere with the analyte’s final elution from the column.
It goes (almost) without saying that mobile phase generation should always make use of the highest purity water, solvents and buffers available, because each of these components contributes to background noise. If a mass spectrogram features a high level of background noise, limits of detection and quantitation will be compromised.
How are samples prepared for injection into liquid chromatography MS?
Solid phase extraction (SPE): This separation technique is often used to separate compounds (e.g., pesticides) dissolved in solutions (e.g., water). A cartridge is packed with an octadecyl (C-18) stationary (chromatographic) phase, which may be silica-based, and it is through this column that the solution is passed. Alternately, a polymer-based cartridge may be utilized if the sample is highly acidic.

Elution of individual compounds occurs over time, and one SPE column can eventually elute several compounds. Eluents can then be concentrated to dryness, diluted with 50:50 methanol/water, and injected into the mass spectrometer. Alternately, SPE can be performed in high-throughput 96-well plates, such as the SOLAµ SPE Plates.
Liquid-liquid extraction (LLE): In LLE, the analyte of interest is first partitioned by a solvent, after which it is extracted, concentrated, and diluted. Phenol-chloroform extraction is one example of an LLE that has historically been used to separate not only protein, but nucleic acids as well. A specific analyte, such as testosterone, is quantitated in samples by first adding the organic solvent methyl-tert-butyl-ether, followed by vortexing. The supernatant is then removed and transferred to a heating block. Following evaporation, the residue is reconstituted with a 50:50 methanol:water solution containing ammonium acetate and formic acid.
Solid supported liquid-liquid extraction (SLE): In SLE, the aqueous sample is loaded onto a support consisting of diatomaceous earth. The sample is allowed to absorb into the support, after which it is washed several times with an organic extraction solvent such as methyl tert-butyl ether. After the analyte has been partitioned into the organic phase, it is concentrated by drying before being reconstituted in a 50:50 methanol:water solution.
Protein precipitation (PPE): Several methods of protein precipitation exist, including desalting, isoelectric point precipitation, organic solvent extraction and ion exchange. The specific chromatography method used depends on sample type (cell, tissue or fluid), location, abundance and size of the target protein, matrix effects, etc.
Desalting: Also called ammonium sulfate precipitation, this protein precipitation technique relies on increasing the concentration of ammonium sulfate until the protein is “salted out.” This process occurs as the salt eventually outcompetes the protein for available water.
Isoelectric point precipitation: Protein solubility is a result of protein ionic strength (pH); if that strength is weakened by a solvent, for example, and the protein’s isoelectric point reaches a net charge of zero, the protein will tend to aggregate and clump.

Organic solvent extraction: Protein solubility often depends on an environment having a large dielectric constant, which is present in solvents such as water. Organic solvents such as acetone and methanol, on the other hand, have small dielectric constants, and so promote the aggregation of protein for mass spec analysis.
During organic solvent extraction, a target protein is precipitated due to its unequal solubility in water and the organic solvent. Following precipitation and concentration, a buffer solution is added to optimize pH. The target protein is then extracted using a solvent such as ether or chloroform.
Ion exchange chromatography: This technique may be used when solvent extraction is encumbered by emulsification and relies on“stopping” proteins at their different isoelectric points. Ion exchange columns use a polymer (e.g., anion-exchange resin DEAE cellulose) and solutions to create a pH gradient. Proteins migrate through this gradient until they reach their isoelectric point, at which point they stop. The bound proteins are eluted by changing the pH or increasing the salt concentration of the elution buffer.
Specific analytes that are routinely analyzed via LC/MS have unique preparation protocols as outlined below:
- Pesticides sample preparation
Understand how to perform sample collection, processing, extraction and clean-up techniques prior to LC and LC/MS analysis. - Protein sample preparation
Learn about the extraction and processing of LC/MS samples such as cells, tissues, serum, plasma and biofluids for proteomics research and drug discovery. - Food sample preparation
Food samples range from breads to oils to dietary supplements. Discover the different protocols required to transform basic food samples to analytical samples ready for LC/MS. - Beverage sample preparation
Beverages such as wine, coffee, tea and water undergo routine analysis for contamination and impurities. Learn how to prepare beverage samples here. - Pharma and Biopharma sample preparation
Characterization of antibodies, glycans and peptides for biotherapeutics requires careful sample preparation prior to screening. Learn more about the process here.
For more information
Liquid chromatography column overview
Liquid chromatography columns are typically offered in HPLC and UHPLC formats. More information about chromatography columns is provided in our Chromatography Learning Center.
The columns themselves consist of two parts: the support matrix (often composed of silica) and the bonded phase (a chemical moiety attached to the silica). Differences in the support matrix determine whether acidic or basic analytes are retained by the column via interactions with different bonded groups. The figure below portrays an example silanol bonded group.
The following materials are frequently used within the column support matrices:
- Monolithic silica: Often used with food, plasma, viscous or “dirty” sample preps, this material contains silica pores that are 50 nm or greater in diameter as well as smaller silica pores that range from 2-50 nm in diameter. The material itself is composed of a single-piece permeable mass rather than individual particles (1). The varied range of pore diameters in this single mass enables fast analysis of samples even when subjected to low back pressures. Monolithic silica is preferentially used to separate large molecules.
- Core-shell: This support material, which is also termed solid core, consists of a solid silica core surrounded by a porous shell. Because the range of particle diameters is much tighter than that of monolithic silica, chromatographic separations are faster and more efficient.

- Traditional silica: Also called fully porous silica, traditional silica columns have extensive surface area and high mechanical strength; such columns are often used for polar samples and are resilient to high back pressures. While traditional silica is prone to hydrolysis at high pH ranges (>pH 7.5), it can be modified by the addition of an organo group to create an organosilica hybrid. Traditional silica can also be modified and function as support material for reversed-phase liquid chromatography (RPLC).
- Porous graphite carbon: Composed of flat sheets of hexagonally arranged carbon atoms, porous graphite carbon contains no interacting silanol groups and can be used to separate samples at extreme pH levels. Porous graphite carbon is often used in RPLC and hydrophilic interaction chromatography (HILIC).
- HILIC mode separation utilizes acetonitrile with a small amount of water or alcohol to create a liquid/liquid extraction system. Salts such as sodium perchlorate may also be added to increase elution capability. HILIC mode separation is often used for metabolic and glycosylation studies.
- Svec F, Huber CG. 2006 Monolithic Materials: Promises, Challenges, Achievements. Anal Chem 78(7):2100–2107.
Featured resources
Chromatography resources
Troubleshoot experimental setup issues and select sample prep protocols and column chemistries for your application.
Sample workflows
Understand recent UHPLC advances, the basics of glycan analysis, and intact antibody characterization.
Webinar
Learn about column chemistries, substrates and formats used for the analysis of hydrophilic anions and cations.
Video
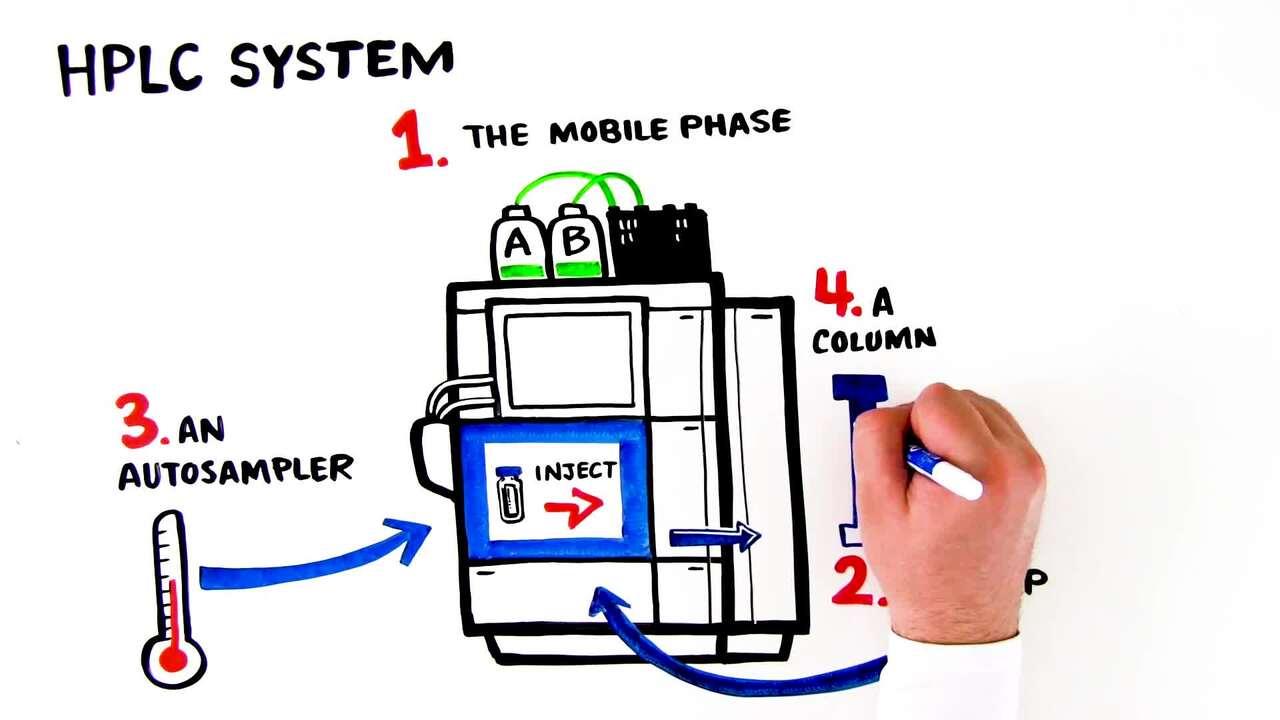
Learn about the basics of HPLC and UHPLC in this short tutorial video.
Mass Spectrometry Resource Library
Access our extensive collection of mass spectrometry workflows, scientific applications, technical handbooks, webinars and more.