Search


Overview
Hydrogen deuterium exchange (HDX) mass spectrometry (MS) is a powerful tool for studying the dynamics of higher order structure of protein-based therapeutics. The rate of hydrogen-to-deuterium exchange within the amide hydrogen on the backbone of biotherapeutics provides solvent accessibility information, and thus protein structure and conformation can be inferred.
Thermo Scientific Orbitrap-based HDX MS offers a robust method for the analysis of protein conformation, conformation dynamics, and protein-protein interactions.
Full structural characterization is critical in biopharmaceutical development where the subtle but critical local conformational changes can impact safety and efficacy.
Key technologies
HDX-MS workflow
Thermo Scientific workflow for hydrogen deuterium exchange mass spectrometry
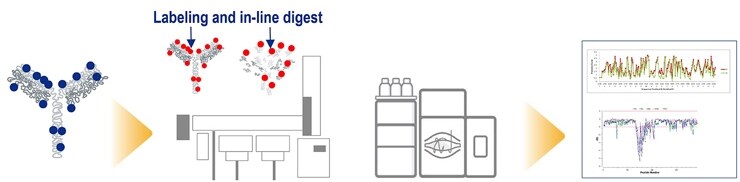
Bottom-up HDX-MS analysis
The most commonly used strategy for HDX-MS is to digest the proteins into peptides and analyze them using mass spectrometry. This ensures complete sequence coverage and captures region-specific information from the protein. Before hydrogen-deuterium exchange is performed, the protein is digested and analyzed in a data-dependent fashion using multiple fragmentation techniques: collision-induced dissociation [CID], higher-energy collisional dissociation [HCD] and electron transfer dissociation [ETD].
Currently available commercial platforms, such as the TRAJAN CHRONECT system, enable automated labelling and digestion.
Intact/top-down HDX-MS analysis
The alternative to bottom-up HDX-MS is intact/top-down analysis. In intact/top-down HDX-MS, proteins are introduced into the mass spectrometer after deuterium exchange without any digestion.
Recommended Products for HDX-MS

H/D-X PAL Hydrogen Deuterium Exchange Sampler system
TRAJAN CHRONECT
The new TRAJAN CHRONECT extended parallel system can accurately schedule fast deuterium exchange time points (approximately 15 seconds) to probe protein conformation dynamics. The TRAJAN CHRONECT autosampler includes:
- A standalone enzyme column
- A chamber to perform on-line peptide digestion
- A closed chamber for stable temperatures for all vials
A flexible 3-valve configuration in the cooling chamber allows efficient sample clean-up. The updated three valve system could set up the back-flash function to efficiently reduce the carryover and clean up the build-up on top of the column which results in very reproducible chromatography. Chronos software provides full editing capabilities for method customization and full integration of Thermo Scientific Xcalibur software.
HPLC Columns

Trap column
- Thermo Scientific Acclaim PepMap 100 C18 HPLC Column (P/N 160434)
- Thermo Scientific PepMap Neo Trap Cartridge Holder (P/N 174502)
- 2 Thermo Scientific nanoViper Fingertight Fittings, 100 µm (P/N 6041.5811)
Generate high-resolution analyses of tryptic, natural, and synthetic peptides using Acclaim PepMap HPLC columns. Due to their high loading capacity, these columns are exceptionally suitable for the analysis of low abundant peptides in complex samples.

Analytical column
- Thermo Scientific HypersilGOLD C18 HPLC Column (50x1 mm, 1.9 um) (P/N 25002-051030). Requires 75 µm tubing inside the cool box; 75 µm tubing after the column.
Achieve exceptional peak shape and resolution for your LC/MS applications with Hypersil GOLD HPLC columns. These endcapped, ultrapure, silica-based columns deliver significant reduction in peak tailing using generic gradients with C18 selectivity. With their excellent resolution, efficiency, and sensitivity, Hypersil GOLD columns give you confidence in the accuracy and quality of your analytical data.
HPLC system
Vanquish Neo UHPLC system
Pursue the next scientific breakthrough with the best performance, productivity, and usability in a nano-, capillary-, and micro-flow UHPLC systems. The Vanquish Neo UHPLC system combines an unrivaled degree of innovation to deliver 24/7 reproducible separations of complex mixtures at maximum performance for a variety of high-sensitivity LC-MS workflow.
Orbitrap Exploris 240 Mass Spectrometers
Deliver on your objectives by combining application flexibility with superior performance for HDX-MS to drive discovery and identification.

Orbitrap Exploris 480 Mass Spectrometer
The high resolution-accurate mass (HRAM) necessary for specificity is available with Orbitrap Exploris 480 Mass Spectrometer, plus short chromatographic runs required to prevent back exchange and to allow precise measurement of deuterium incorporation. High quality Orbitrap DDA spectra in conjunction with Thermo Scientific BioPharma Finder Software ensures confident and rapid identification of all peptides
Orbitrap Ascend Structural Biology Tribrid Mass Spectrometer
Built to meet the demands of structural biology, the Orbitrap Ascend Structural Biology Tribrid mass spectrometer adds innovations to scale up characterization of biomolecular structures, including protein-protein or protein-ligand interactions on intact level or peptide-level.
Orbitrap Eclipse Tribrid Mass Spectrometer
Delivering the ultimate flexibility to expand experimental scope, and with built-in intelligence, Orbitrap Eclipse Tribrid Mass Spectrometer ensures the highest data quality for HDX-MS experiments. It delivers the high resolution-accurate mass necessary for specificity with short chromatographic runs required to prevent back exchange and to allow precise measurement of deuterium incorporation. Multiple fragmentation techniques, CID, HCD, UVPD and ETD are available to identify as many overlapping peptides as possible, enabling maximum sequence coverage for protein identification. Plus, it offers ultimate precision with ETD or UVPD fragmentation to allow localization of deuterium exchange to the amino acid level.
BioPharma Finder Software
Thermo Scientific BioPharma Finder software supports all HDX-MS data analysis, including peptide identification, PTM analysis and HDX-unique protection factor plots at the single residue level.
Resources
HDX-MS for the Masses
HDX-MS Higher Order Protein Structure Elucidation
Structural Characterization of a Therapeutic Antibody & Biosimilar
HDX-MS Protein Structural Characterization
Hydrogen Deuterium Exchange (HDX) Mass Spectrometry Learning Centre
Featured webinars
Running the Gamut - Tales of Protein Dynamics in Drug Discovery,
Benjamin T. Walters. Scientist, Genentech
Application of HDX-MS for Drug Discovery & Development
Pfizer & BioAnalytix: Improved Biotherapeutic Characterization using HDX-M
No records were found matching your criteria
Contact us
* Required field














