Search
PI3K Signaling in B-Lymphocytes
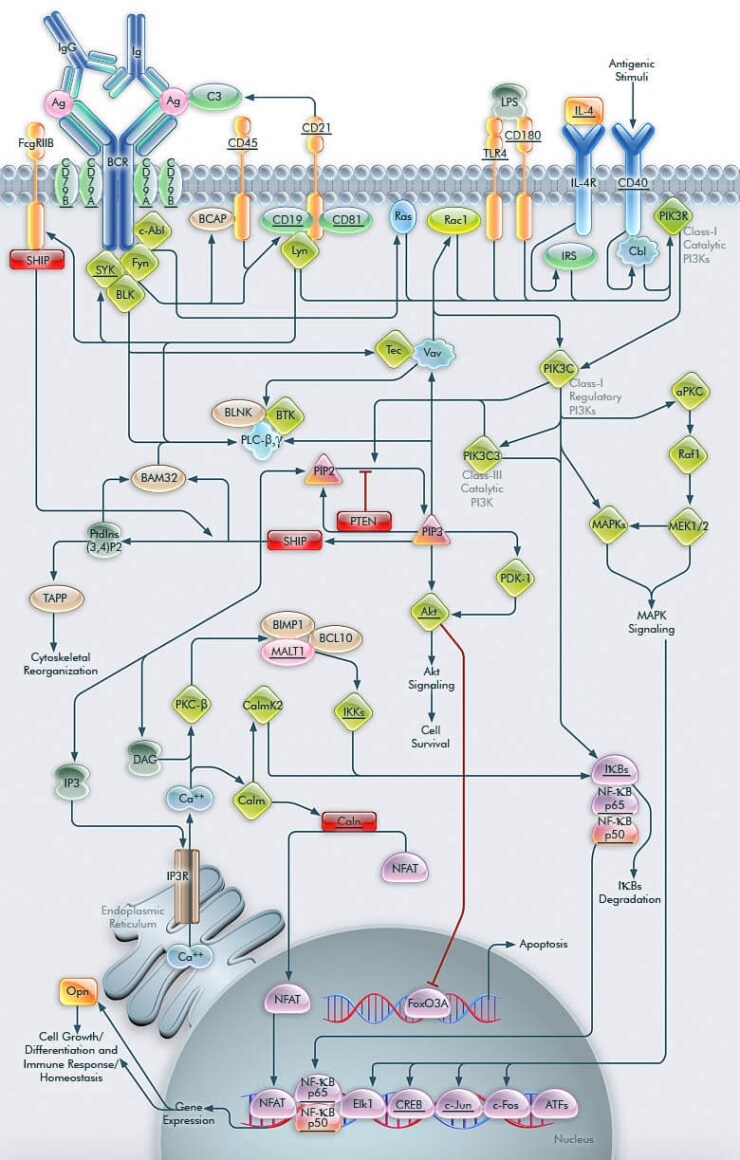
PI3Ks (Phosphoinositide-3-Kinases) regulate numerous biological processes, including cell growth, differentiation, survival, proliferation, migration, and metabolism. In the immune system, impaired PI3K signaling leads to immunodeficiency, whereas unrestrained PI3K signaling contributes to autoimmunity and leukemia. The Class I and III PI3Ks basically facilitate B cell development through defined stages, resulting in at least three distinct lineages of mature B-lymphocytes. In B cells, PI3K is activated within seconds of antigen-receptor triggering. The BCR (B-cell antigen receptor) plays a critical role in recognition of antigens and activation of B cells. The BCR or mIg (membrane immunoglobulin) is associated with Ig-Alpha/CD79A (CD79A antigen) and Ig-Beta/CD79B (CD79B antigen) heterodimer. The mIg subunits bind antigen and cause receptor aggregation, while the Ig-Alpha/Ig-Beta subunits transduce signals to the cell interior [1,2]. Engagement of the BCR-antigen complex activates intracellular protein tyrosine kinases like SYK (Spleen Tyrosine Kinase), BTK (Bruton Agammaglobulinemia Tyrosine Kinase), and Fyn (Fyn Oncogene Related to Src, FGR, YES). These tyrosine kinases phosphorylate the co-receptors CD19 (CD19 Antigen) and BCAP (B-Cell Adaptor Protein) at the YXXM motifs, which provide binding sites for PI3Ks. CD19 is one of the main regulators of PI3K activity in B cells. CD19 has an important, but not indispensable, role in PI3K activation; rather, it is required for sustained PI3K activation after BCR stimulation. The co-receptor complex is also composed of CD21 (CD21 Antigen) and CD81. CD21 binds opsinized antigenic particles and activate Complement Component-C3, a reaction central to complement function in the immune response and sustained BCR signaling. Many other transmembrane receptors are known to modulate specific elements of BCR signaling. A few of these are CD45 (CD45 Antigen) and Fc-GammaRIIB (Low Affinity Immunoglobulin Gamma Fc Region Receptor-IIB). CD45 occurs as a component of a complex of proteins associated with the antigen receptor, and may regulate signal transduction by modulating the phosphorylation state of the co-receptors like CD19 [2,3].
Moreover for B-cell development, the capacity of CD19 to promote thymus-dependent immune responses is linked to its capacity to recruit and activate PI3K. CD19 phosphorylation activates Lyn (V-YES-1 Yamaguchi Sarcoma Viral Related Oncogene Homolog) that in turn recruits regulatory PI3Ks. Lyn activation stimulates BCR and associated protein tyrosine kinases to activate Vav (Oncogene Vav) and Tec (Tec Protein Tyrosine Kinase). Vav and Tec contribute to PI3K activation in B-cells by a mechanism that involves the activation of Rac1 (Ras-Related C3 Botulinum Toxin Substrate-1), which then binds to regulatory PI3Ks through its RhoGAP (Rho-GTPase-Activating Protein) domain. This mechanism places Vav and Tec upstream of PI3K signaling. B-cell proliferation is also stimulated by IL-4 (Interleukin-4)/IL-4R (Interleukin-4 Receptor) through IRS (Insulin Receptor Substrate) activation; LPS (Lipopolysaccharide) stimulated TLR4 (Toll-Like Receptor-4)/CD180 (CD180 Antigen) activation; or a CD40 (B-Cell-Associated Molecule CD40) activated Cbl (Cas-Br-M (Murine) Ecotropic Retroviral Transforming Sequence) that engages p85Alpha-associated p110Delta, thus enhancing PI3K signaling. The regulatory Class I PI3K subunits then activate subsequent catalytic Class I subunits [4,5]. Once active, the catalytic Class I PI3Ks phosphorylate PIP2 (Phosphatidylinositol 4,5-Bisphosphate) to produce PIP3 (Phosphatidylinositol-3,4,5-trisphosphate) at the inner leaflet of the plasma membrane and also activate the Class III PIK3C3 (Phosphoinositide-3-Kinase-Class-3) to enhance the phosphorylation process. PI3K/PIP3 signaling then turns on Akt Signaling via Akt (v-Akt Murine Thymoma Viral Oncogene Homolog)/PDK-1 (Phosphoinositide-Dependent Kinase-1) activation and down regulation of apoptosis regulating Forkhead family of transcription factors like FoxO3A (Forkhead Box-O3A), and thereby facilitates cell survival. However, the conversion of PIP2 to PIP3 is reversed by PTEN (Phosphatase and Tensin Homolog), a lipid phosphatase, which removes the D3 phosphate from PIP3 [6]. A similar inhibitory function is also exhibited by the Fc-Gamma-RIIB induced SHIP (SH2-Containing Inositol Phosphatase). Upon BCR co-ligation, Lyn and associated co-receptors generate an SH2 recognition domain that provides the binding site for the inhibitory signaling molecule SHIP, which removes excess accumulation of PIP3 by converting it to PtdIns(3,4)P2 (Phosphatidylinositol 3,4-Bisphosphate). This leads to the abrogation of PI3K/PIP3 signaling. Thus the PI3K signal is taken over by TAPP (Tandem PH-Domain-Containing Protein) adaptor proteins, which have binding specificity for PtdIns(3,4)P2 and influence the process of cytoskeletal reorganization [7,8].
The conversion of PIP3 to PtdIns(3,4)P2 also terminates activation of Vav, Tec and the Signalosome complex that comprises of BTK (Bruton Agammaglobulinemia Tyrosine Kinase), PLC-Beta (Phospholipase-C-Beta), PLC-Gamma (Phospholipase-C-Gamma), and BLNK (B-Cell Linker Protein). Generation of PIP3 and PtdIns(3,4)P2 activates BAM32 (B-Cell Adaptor Molecule-32KD), an adaptor protein with a high affinity PIP3-binding PH domain that generates BCR-dependent Calcium flux. After BCR ligation, BAM32 is recruited to the plasma membrane through its PH domain. Moreover, BCR-stimulated calcium flux also depends on the Signalosome activation by BCR-associated co-receptors, BAM32, Vav, and Tec. Sequential phosphorylation by Lyn and autophosphorylation of BTK contributes to the BCR-stimulated and PI3K-dependent calcium signaling, by phosphorylating and activating PLC-Beta and Gamma. This process is also well regulated by BAM32. These Phospholipases utilize PIP2 as a substrate for the generation of IP3 (Inositol-1,4,5-trisphosphate) and DAG (Diacylglycerol). IP3 activates IP3R (IP3 Receptor) to release stored calcium ions, whereas DAG and calcium activate PKC-Beta (Protein Kinase-C-Beta) and Calm (Calmodulin), respectively. One of the targets regulated by calcium elevation is the transcription factor NFAT (Nuclear-Factor of Activated T-cells), whose nuclear translocation is facilitated through its dephosphorylation by Caln (Calcineurin) by Calm. PKC-Beta appears to phosphorylate IKKs (I-KappaB Kinases) activity resulting in the phosphorylation and degradation of I-KappaBs (Inhibitor of Kappa Light Chain Gene Enhancer in B-Cells) and translocation of NF-KappaB (Nuclear Factor-Kappa-B) to the nucleus. PKC activates BIMP1 (Bcl10-Interacting MAGUK Protein-1)/Bcl10 (B-Cell Cell/Lymphoma-10)/MALT1 (Mucosa Associated Lymphoid Tissue Lymphoma Translocation Gene-1) that forms a strong and specific complex within the cell to synergize in the activation of NF-KappaB, whereas, Calm activates CaMKII (Calcium/Calmodulin-Dependent Protein Kinase-II) to facilitate the same [9,10]. Translocation of NF-KappaB by catalytic PI3Ks further enhances the BCR-stimulated PI3K gene expression. PI3K mediated gene expression under the influence of BCR/c-Abl activate the MAPK cascade via aPKC (Atypical PKC)/Raf1 (v-Raf1 Murine Leukemia Viral Oncogene Homolog-1)/MEK (MEK Kinase) route. Further activation of MAPKs regulate subsequent MAPK pathways to control cell proliferation through induction of transcription factors [like NF-KappaB, NFAT, Elk1 (ETS-domain protein Elk1), ATFs (Activating Transcription Factors), and CREB (cAMP Response Element-Binding Protein)]. These factors also regulate Opn (Osteopontin) gene expression that modulates cellular processes like immune homeostasis, ossification, and myelination. Upon translocation to the exterior, Opn controls the integrin pathway through ECM/Integrin interactions to recruit the regulatory PI3Ks [11].
The production of second messenger by PI3K and activation of intermediate signaling proteins is the concerted regulation of several transcription factors that mediate gene transcription in the B cells. Since the immune response in vivo is regulated by alteration of these signaling outcomes, achieving a precise understanding of intracellular molecular events leading to B-lymphocyte proliferation, deletion, receptor editing, and survival still remains a challenge for the future. Understanding the contributions of PI3Ks to lymphocyte biology may contribute to the development of treatments for immune-related diseases, including autoimmunity, leukemia, and graft rejection [12].
Pathway
Key

抗体资源库
有针对性地收集科学应用说明、方法和细胞信号图。
Related products
- Al-Alwan MM, Okkenhaug K, Vanhaesebroeck B, et al. (2007) Requirement for phosphoinositide 3-kinase p110delta signaling in B cell antigen receptor-mediated antigen presentation. J Immunol 178(4):2328-35.
- Beer-Hammer S, Zebedin E, von Holleben M, et al. (2010) The catalytic PI3K isoforms p110gamma and p110delta contribute to B cell development and maintenance, transformation, and proliferation. J Leukoc Biol 87(6):1083-95. Epub 2010 Mar 3.
- Zhang TT, Li H, Cheung SM, et al. (2009) Phosphoinositide 3-kinase-regulated adapters in lymphocyte activation. Immunol Rev 232(1):255-72. Review.
- Hebeis B, Vigorito E, Kovesdi D, et al. (2005) Vav proteins are required for B-lymphocyte responses to LPS. Blood 106(2):635-40. Epub 2005 Apr 5.
- Deambrosis I, Scalabrino E, Deregibus MC, et al. (2005) CD40-dependent activation of phosphatidylinositol 3-kinase/AKT pathway inhibits apoptosis of human cultured mesangial cells induced by oxidized LDL. Int J Immunopathol Pharmacol 18(2):327-37.
- Tybulewicz VL (2004) Commentary: New insights into the complexity of phosphatidylinositol lipid signaling in B lymphocytes. Eur J Immunol 34(11):2964-7.
- Decker SJ, Saltiel AR (2005) Staying in SHIP shape. Nat Med 11(2):123-4.
- Costantini JL, Cheung SM, Hou S, et al. (2009) TAPP2 links phosphoinositide 3-kinase signaling to B-cell adhesion through interaction with the cytoskeletal protein utrophin: expression of a novel cell adhesion-promoting complex in B-cell leukemia. Blood 114(21):4703-12. Epub 2009 Sep 28.
- Hess KL, Donahue AC, Ng KL, et al. (2004) Frontline: The p85alpha isoform of phosphoinositide 3-kinase is essential for a subset of B cell receptor-initiated signaling responses. Eur J Immunol 34(11):2968-76.
- Hebeis BJ, Vigorito E, Turner M (2004) The p110delta subunit of phosphoinositide 3-kinase is required for the lipopolysaccharide response of mouse B cells. Biochem Soc Trans 32(Pt 5):789-91. Review.
- Kharas MG, Fruman DA (2005) ABL oncogenes and phosphoinositide 3-kinase: mechanism of activation and downstream effectors. Cancer Res 65(6):2047-53. Review.
- Stauffer F, Holzer P, García-Echeverría C (2005) Blocking the PI3K/PKB pathway in tumor cells. Curr Med Chem Anticancer Agents 5(5):449-62. Review.
