Search

Endocrinology and Metabolism ELISA Kits |
Measure hormonal and metabolic markers
Thermo Fisher Scientific offers a comprehensive selection of highly sensitive ELISA and multiplex immunoassay solutions for studying steroids, hormones, peptides, and metabolites in a variety of biological matrices. Our endocrinology and metabolic hormone immunoassay portfolio enable sensitive, reliable detection of a broad spectrum of targets using complementary technologies, including ELISA, proximity ligation assays (PLA), and multiplex immunoassays. These platforms support research across diverse applications—from focused single-analyte quantification to high-throughput profiling.
By combining assay sensitivity, flexibility, and breadth of coverage, our immunoassay portfolio enables deeper insight into the complex hormonal networks—supporting discovery across physiology, toxicology, and disease research.
Overview of endocrinology and metabolic markers
Endocrinology and metabolism are fundamental areas of life science research focused on understanding how the endocrine system maintains physiological balance through hormone signaling. The endocrine system consists of specialized glands—such as the pituitary, thyroid, adrenal, pancreas, and gonads—that secrete hormones directly into the bloodstream and travel to target organs to coordinate essential functions (Figure 1).
But what are hormones? Hormones are biochemical messengers and are fundamental regulators of physiological balance, orchestrating communication between the nervous system, endocrine organs, and metabolic tissues. From neuropeptides that shape brain signaling to steroid, thyroid, and pituitary hormones that govern growth, reproduction, stress response, and energy homeostasis, precise hormone measurement is essential for understanding both normal physiology and disease mechanisms.
Understanding what hormones are and how they function is foundational to endocrinology. Hormones bind to specific receptors on or inside target cells to modulate activity, influencing energy utilization, stress responses, development, mood, and metabolic balance. Disruptions in hormonal signaling can lead to metabolic disorders such as diabetes, thyroid dysfunction, or metabolic syndrome, making endocrinology critical to both basic biology and clinical research. [1]

Furthermore, hormone signaling extends far beyond traditional glands and involves dynamic interactions between multiple systems of the body. Described as the neuroendocrine system, this system plays a crucial role in regulating immune function and broader physiological responses (Figure 2). It shows that hormonal and neurochemical pathways are deeply integrated, with signals from the brain influencing immune regulation and systemic homeostasis, and vice versa. This interplay emphasizes that endocrine communication is not isolated to discrete glands but is part of a complex, body-wide network of hormone-mediated regulation that impacts metabolism, stress responses, immune function, and disease susceptibility. [2,3]
In research settings, endocrinology provides important insight into normal physiology and disease pathophysiology. Dysregulation of hormone production, secretion, or receptor signaling is associated with a wide range of conditions, including diabetes, metabolic syndrome, obesity, thyroid disorders, infertility, cancer, and neuroendocrine diseases. As a result, hormone testing is a foundational component of endocrinology and metabolism research, enabling scientists to investigate signaling pathways, identify biomarkers, and evaluate the biological impact of experimental therapies.
Because hormones can be present across a wide range of concentrations and exhibit dynamic, tightly regulated expression patterns, accurate, and sensitive measurement is critical. Immunoassays are widely used in endocrinology research due to their high sensitivity, specificity, and compatibility with diverse sample types. These assays enable reliable quantification of endocrine markers, supporting reproducible data generation and robust interpretation of experimental results.
Key targets of the pituitary gland, which secretes hormones that act on peripheral endocrine organs:
- Adrenocorticotropic hormone (ACTH)—stimulates cortisol production in the adrenal cortex.
- Thyroid-stimulating hormone (TSH)—regulates thyroid hormone synthesis and secretion. It is a highly sensitive marker of thyroid metabolic activity and is central to diagnosing hypo- and hyperthyroidism.
- Luteinizing hormone (LH) and follicle-stimulating hormone (FSH)—regulate gonadal function. LH stimulates testosterone production in testes and ovulation and progesterone synthesis in ovaries. FSH supports spermatogenesis and follicular development. It indirectly affects metabolism through its regulation of sex steroids.
- Growth hormone (GH)—promotes lipolysis, increases protein synthesis, and reduces glucose uptake, shifting metabolism toward fat utilization.
- Prolactin—supports lactation and modulates immune and reproductive function. Elevated prolactin suppresses GnRH, leading to infertility and metabolic disturbances.
- Oxytocin—regulates uterine contractions, lactation, and social bonding, with emerging roles in appetite and glucose metabolism.
Key targets of the thyroid gland, which regulates metabolism, thermogenesis, and development:
- Thyroxine (T4)—the primary hormone produced by the thyroid which serves as a prohormone converted to T3 in peripheral tissues. It reflects overall thyroid output.
- Triiodothyronine (T3)—the biologically active thyroid hormone. It increases mitochondrial activity, oxygen consumption, thermogenesis, and lipid and carbohydrate metabolism.
- Thyroglobulin—a precursor protein for thyroid hormone synthesis which is used as a tumor marker in differentiated thyroid cancer.
- Thyroid peroxidase antibodies (TPO-Ab)—indicate autoimmune thyroid destruction, which leads to hypothyroidism and metabolic slowing.
Key targets of the pancreas (endocrine), which is central to glucose and energy metabolism:
- Insulin—the key anabolic hormone that promotes glucose uptake, glycogen synthesis, lipogenesis, and protein synthesis. Insulin deficiency or resistance underlies diabetes.
- C-Peptide—reflects endogenous insulin production and beta-cell function, useful in distinguishing between diabetes types.
- Glucagon—counteracts insulin by stimulating glycogen breakdown and gluconeogenesis during fasting.
- Amylin—slows gastric emptying and suppresses glucagon after meals, supporting postprandial glucose control.
- HbA1c—reflects average glucose exposure over ~3 months and integrates long-term metabolic control.
These glands are central hubs for hormone synthesis and release. However, modern research has repeatedly shown that hormone production is not exclusive to glands. Many other tissues and organs also produce hormones that regulate metabolism, appetite, immune function, growth, and more [4].
A particularly well-studied example comes from adipose tissue (body fat). Once considered a passive energy store, adipose tissue is now recognized as a biologically active endocrine organ. Fat cells (adipocytes) secrete hormones—often called adipokines—such as leptin and adiponectin, that circulate in the bloodstream and directly influence energy balance, appetite, insulin sensitivity, and metabolic health. [5]
- Leptin, produced in proportion to fat mass, acts on the hypothalamus to reduce food intake and modulate energy expenditure, linking nutritional state to neuroendocrine control of appetite.
- Adiponectin, also derived from adipose tissue, plays an important role in enhancing insulin sensitivity and protecting against inflammation and vascular dysfunction.
Other tissues similarly contribute to endocrine signals. For example, cells in the gastrointestinal tract release hormones like ghrelin that stimulate appetite and influence metabolic processes, while the kidneys, heart, skin, and placenta secrete hormones involved in blood pressure regulation, calcium balance, immune modulation, and pregnancy physiology.
Endocrinology and metabolism ELISA kits
Endocrinology and metabolism ELISA kits enable sensitive, quantitative measurement of hormones and metabolic biomarkers in serum, plasma, cell culture supernatants, and other sample types—supporting research in diabetes, obesity, thyroid function, stress biology, and related pathways.
For example, GLP-1 (glucagon-like peptide-1) is critically important for research because it sits at the intersection of glucose metabolism, appetite regulation, cardiovascular biology, and therapeutic innovation. It helps provide a unifying biological pathway linking type 2 diabetes, obesity, and cardiometabolic disease.
Linearity-of-dilution data and the standard curve of Human GLP-1 using the Human GLP-1 ELISA Kit are shown in Figure 3 and Figure 4.
Search endocrinology ELISA kits Search metabolic kits
Learn more about ELISA kits and components
Popular endocrine and metabolic protein targets and ELISA performance data
Table 1. View our kits for the listed popular targets.
Table 2. List of publications highlighting Invitrogen ELISA kits used in endocrinology and metabolic research.
| Reference | Publication title | Target/s | Testing method |
|---|---|---|---|
| Gurevich et al. 2020. | iPSC-derived hepatocytes generated from NASH donors provide a valuable platform for disease modeling and drug discovery | Albumin | Human Albumin ELISA Kit |
| Ma et al. 2022. | Sennoside A Induces GLP-1 Secretion Through Activation of the ERK1/2 Pathway in L-Cells | GLP-1 | Human GLP-1 ELISA Kit |
| Dargam et al. 2025. | Phosphate salt selection affects mortality and vascular calcification in the adenine-induced chronic kidney disease mouse model | Urea nitrogen (BUN) | Urea Nitrogen (BUN) Colorimetric Detection Kit |
| Chen et al. 2021. | Decreased blood vessel density and endothelial cell subset dynamics during ageing of the endocrine system | Thyroxine (T4) | Thyroxine (T4) Competitive ELISA Kit |
| Stratton et al. 2020. | Four Weeks of Time-Restricted Feeding Combined with Resistance Training Does Not Differentially Influence Measures of Body Composition, Muscle Performance, Resting Energy Expenditure, and Blood Biomarkers | Cortisol | Human Cortisol Competitive ELISA Kit |
Endocrinology and metabolism ProQuantum high sensitivity immunoassays
Invitrogen ProQuantum high-sensitivity immunoassays are designed to help provide a high-performance quantitative protein measurement from 2–5 µL of sample. The assay is fast and easy to run, with no proprietary instrument to purchase. Utilizing proximity-based amplification technology, ProQuantum assays offer the analyte specificity of high-affinity antibody-antigen binding with the signal detection and amplification capabilities of real-time PCR to achieve a highly sensitive protein quantitation assay. ProQuantum assays can typically detect lower levels of protein with lower sample consumption than traditional methods and offer a broader dynamic range. This is important to accurately detect variations in concentration of endocrine and metabolic markers as they can vary depending on the metabolic state and disease state, as well as time of day.
Find endocrinology-related ProQuantum assays
Learn more about how the ProQuantum immunoassays work
Read BioProbes Journal article: Introducing ProQuantum High-Sensitivity Immunoassays—The new generation of target-specific protein quantitation
Table 3. View our ProQuantum immunoassays for the following popular targets.
Endocrinology and metabolism ProcartaPlex multiplex immunoassays
Invitrogen ProcartaPlex multiplex immunoassays help provide a powerful platform for the simultaneous quantification of up to 9 endocrine and metabolic biomarkers, supporting researchers in illuminating the reasons for endocrine dysregulation, metabolic changes, and complex feedback mechanisms across multiple organ systems. ProcartaPlex assays demonstrate strong correlation to traditional ELISA methods, supporting confidence in data comparability while enabling multiplex efficiency (Figure 6).
Hormones can be present across a wide range of concentrations. ProcartaPlex assays are designed with a broad dynamic range, allowing accurate detection of these varying levels within a single run. This enables reliable measurement of hormones, verified in cerebrospinal fluid (CSF), serum, plasma, and cell culture supernatant samples. Other biological samples might be suitable for use in the assays.
As hormones can also influence immunological and neurological processes, the ProcartaPlex platform enables simultaneous measurement of hormones alongside key immune and neurological biomarkers in a single multiplex panel. Because many hormones, particularly steroid and small peptide hormones, are low-molecular-weight targets with limited epitopes, competitive immunoassays are the preferred format for accurate quantification. ProcartaPlex combines competitive assays for small-molecule hormones with sandwich assays for larger proteins such as cytokines and peptide biomarkers, all within the same workflow. This can be exploited by combining several ProcartaPlex Simplex kits into a panel or by custom blended Mix & Match panels. This integrated approach has been extensively tested for cross-compatibility and can provide significant efficiency in time, sample volume, and cost compared with running multiple single-plex assays.
Select from our preconfigured panels described in Table 4 or use the Panel Configurator below to customize your specific panel.
ProcartaPlex Panel Configurator
Learn more about ProcartaPlex multiplex immunoassays
Preconfigured endocrinology multiplex immunoassay panels and performance data
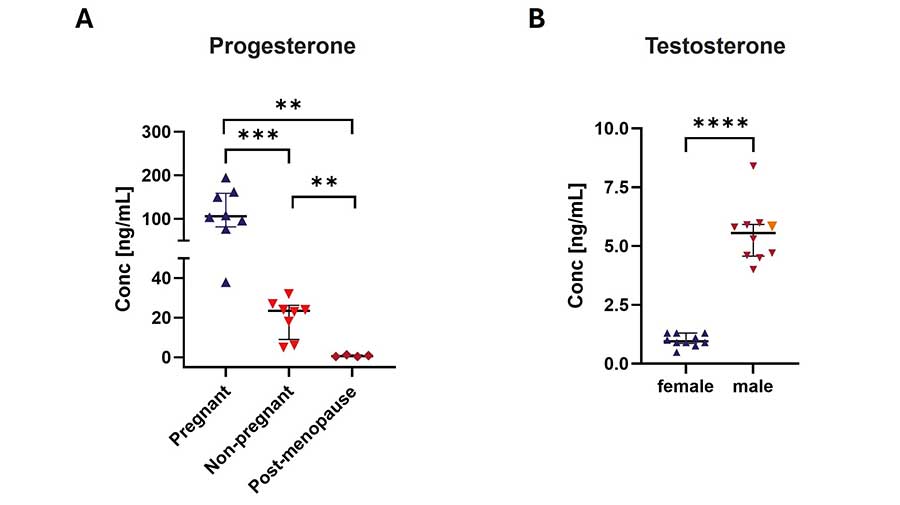
Figure 8A. Progesterone serum levels in females across physiological states. Serum samples from pregnant (n=8), non-pregnant (n=8), and post-menopause (n=4) females were run in duplicates using Invitrogen ProcartaPlex Human Steroid and Thyroid Hormones Panel, 7plex. Compared with age-matched healthy, non-pregnant controls, pregnant females exhibit significantly increased progesterone levels, whereas postmenopausal females show significantly reduced progesterone levels relative to healthy, non-pregnant controls.
Figure 8B. Testosterone serum levels in females and males. Serum samples from female (n=10) and male (n=10) individuals, including NIST® SRM® Standard Reference Material® 971a with a certified concentration value for males (5,838 ng/mL, indicated in orange), were run in duplicates using Invitrogen ProcartaPlex Human Steroid and Thyroid Hormones Panel, 7plex.
Both data are presented as medians with interquartile ranges (IQR). Group differences and statistical significance were assessed by the Mann–Whitney U test (two-tailed); * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001.
Table 4. Preconfigured ProcartaPlex multiplex immunoassay panels for endocrinology.
| Human multiplex hormone panels | ||
|---|---|---|
| Name | Size | Cat. No. |
| ProcartaPlex Human Neuropeptide and Pituitary Hormones Panel, 9plex Target list [bead region]: Neuropeptide Y [42], ACTH [67], Cholecystokinin (CCK) [19], Follicle Stimulating Hormone (FSH) [77], Growth Hormone (GH) [48], Thyroid Stimulating Hormone (TSH) [28], Luteinizing Hormone (LH) [53], Prolactin [22], hCG [56] | 96 tests | EPX090-15861-901 |
| ProcartaPlex Human Steroid- and Thyroid Hormones Panel, 7plex Target list [bead region]: Triiodothyronine (T3) Competitive [38], Thyroxine (T4) Competitive [63], Testosterone Competitive [37], Estradiol (E2) Competitive [26], Progesterone Competitive [35], DHEA-S Competitive [43], Cortisol Competitive [12] | 96 tests | EPX070-15862-901 |
Endocrinology and metabolism QuantiGene multiplex immunoassays
QuantiGene assays are highly sensitive and specific for the detection and quantification of RNA targets. This technology utilizes branched DNA signal amplification to enable the measurement of gene expression levels in a variety of sample types, including blood, tissue, and cell lysates. With up to 80 genes simultaneously measured in a single sample, researchers can focus on endocrinology and hormone markers implicated in biochemical processes. A few examples of published data using QuantiGene panels are highlighted in Table 5.
There are several advantages to bead-based assays over traditional methods of gene expression analysis. QuantiGene assays help provide a highly multiplexed approach, allowing for the simultaneous measurement of multiple targets in a single sample using the same Luminex xMAP technology as ProcartaPlex assays. This capability not only helps save time and resources but also helps provide a more comprehensive understanding of gene expression patterns. The assay is also highly sensitive, enabling the detection of low-abundance targets with high precision.
QuantiGene gene expression assays are versatile tools that can be customized to help meet your specific research needs. Researchers looking to study genes implicated in metabolism or obesity can choose from a selection of preconfigured panels in Table 6. Alternatively, by using the Panel Configurator below, you can choose from a large inventory of verified genes and pathways of interest to create a customized panel for studying endocrinology.
Learn more about QuantiGene multiplex immunoassays
Table 5. List of publications highlighting custom-designed QuantiGene assays for studying endocrinology.
| Reference | Publication summary |
|---|---|
| Ivanova et al., 2024. | A preliminary study was conducted using a custom QuantiGene panel, which was used to investigate the relationship between genetic expression markers in schizophrenic males with metabolic disorders. |
| Baruch, et al., 2023. | The group aimed to characterize Growth Differentiation Factor 15 (GDF15) as a hormone biomarker that is secreted within the central nervous system. A custom QuantiGene plex panel was designed to measure genes of interest in the ISR or Gdf-15 pathways. |
| Bellet et al., 2023. | To determine how much the tryptophan metabolism transcriptional pathway is regulated via the circadian system, mRNA levels of lung and ileum samples of mice were quantified using a custom-designed QuantiGene plex panel. |
| Serradas et al., 2021. | A custom QuantiGene panel was used to investigate the sweet taste transduction pathway and evaluate the hypothesized changes from metabolic diseases, including GLP-1. |
| Beckman et al., 2013. | To explore the endocrine growth axis of coho salmon, a custom QuantiGene plex panel was designed. Liver homogenates were prepared, and the mRNA levels of various hormones and receptors were measured. |
Table 6. Preconfigured QuantiGene gene expression multiplex immunoassay panels for metabolism and obesity research.
| Product Name | Size | Cat. No. |
|---|---|---|
| Human endocrinology panels | ||
| QuantiGene Plex Human Metabolism Panel, 25-plex Target list: PPARG, ADRB2, UCP1, INSR, IRS1, GNPDA2, TBC1D1, FABP4, TNNI3K, TFAP2B, PCSK1, MAF, TMEM160, CLOCK, ADIPOQ, HHEX, PLIN1, SLC6A14, FAIM2, SIRT1, GCG, IAPP, PPIB, TBP, HPRT1 | 1 plate | QGP-125-HUMETAB |
| QuantiGene Plex Human Obesity Panel, 20-plex Target list: MC4R, LEP, LEPR, GLP1-R, GCG (GLP-1), TMEM18, BDNF, KCTD15, SH2B1, MTCH2, NEGR1, SEC16B, FTO, POMC, MC3R, NMB, INHBE, PPIB, TBP, HPRT1 | 1 plate | QGP-120-HUOBESITY |
| Mouse endocrinology panels | ||
| QuantiGene Plex Mouse Metabolism Panel, 25-plex Target list: Pparg, Adrb2, Ucp1, Insr, Irs1, Gnpda2, Tbc1d1, Fabp4, Tnni3k, Tfap2b, Pcsk1, Maf, Tmem160, Clock, Adipoq, Hhex, Plin1, Slc6a14, Faim2, Sirt1, Gcg, Iapp, Tbp, Hprt1, Ppib | 1 plate | QGP-125-MSMETAB |
| QuantiGene Plex Mouse Obesity Panel, 20-plex Target list: Mc4r, Lep, Lepr, Glp1r, Gcg, Tmem18, Bdnf, Kctd15, Sh2b1, Mtch2, Negr1, Sec16b, Fto, Pomc, Mc3r, Nmb, INHBE, PPIB, TBP, HPRT1 | 1 plate | QGP-120-MSOBESITY |
Overview of endocrinology and metabolic markers
Endocrinology and metabolism are fundamental areas of life science research focused on understanding how the endocrine system maintains physiological balance through hormone signaling. The endocrine system consists of specialized glands—such as the pituitary, thyroid, adrenal, pancreas, and gonads—that secrete hormones directly into the bloodstream and travel to target organs to coordinate essential functions (Figure 1).
But what are hormones? Hormones are biochemical messengers and are fundamental regulators of physiological balance, orchestrating communication between the nervous system, endocrine organs, and metabolic tissues. From neuropeptides that shape brain signaling to steroid, thyroid, and pituitary hormones that govern growth, reproduction, stress response, and energy homeostasis, precise hormone measurement is essential for understanding both normal physiology and disease mechanisms.
Understanding what hormones are and how they function is foundational to endocrinology. Hormones bind to specific receptors on or inside target cells to modulate activity, influencing energy utilization, stress responses, development, mood, and metabolic balance. Disruptions in hormonal signaling can lead to metabolic disorders such as diabetes, thyroid dysfunction, or metabolic syndrome, making endocrinology critical to both basic biology and clinical research. [1]

Furthermore, hormone signaling extends far beyond traditional glands and involves dynamic interactions between multiple systems of the body. Described as the neuroendocrine system, this system plays a crucial role in regulating immune function and broader physiological responses (Figure 2). It shows that hormonal and neurochemical pathways are deeply integrated, with signals from the brain influencing immune regulation and systemic homeostasis, and vice versa. This interplay emphasizes that endocrine communication is not isolated to discrete glands but is part of a complex, body-wide network of hormone-mediated regulation that impacts metabolism, stress responses, immune function, and disease susceptibility. [2,3]
In research settings, endocrinology provides important insight into normal physiology and disease pathophysiology. Dysregulation of hormone production, secretion, or receptor signaling is associated with a wide range of conditions, including diabetes, metabolic syndrome, obesity, thyroid disorders, infertility, cancer, and neuroendocrine diseases. As a result, hormone testing is a foundational component of endocrinology and metabolism research, enabling scientists to investigate signaling pathways, identify biomarkers, and evaluate the biological impact of experimental therapies.
Because hormones can be present across a wide range of concentrations and exhibit dynamic, tightly regulated expression patterns, accurate, and sensitive measurement is critical. Immunoassays are widely used in endocrinology research due to their high sensitivity, specificity, and compatibility with diverse sample types. These assays enable reliable quantification of endocrine markers, supporting reproducible data generation and robust interpretation of experimental results.
Key targets of the pituitary gland, which secretes hormones that act on peripheral endocrine organs:
- Adrenocorticotropic hormone (ACTH)—stimulates cortisol production in the adrenal cortex.
- Thyroid-stimulating hormone (TSH)—regulates thyroid hormone synthesis and secretion. It is a highly sensitive marker of thyroid metabolic activity and is central to diagnosing hypo- and hyperthyroidism.
- Luteinizing hormone (LH) and follicle-stimulating hormone (FSH)—regulate gonadal function. LH stimulates testosterone production in testes and ovulation and progesterone synthesis in ovaries. FSH supports spermatogenesis and follicular development. It indirectly affects metabolism through its regulation of sex steroids.
- Growth hormone (GH)—promotes lipolysis, increases protein synthesis, and reduces glucose uptake, shifting metabolism toward fat utilization.
- Prolactin—supports lactation and modulates immune and reproductive function. Elevated prolactin suppresses GnRH, leading to infertility and metabolic disturbances.
- Oxytocin—regulates uterine contractions, lactation, and social bonding, with emerging roles in appetite and glucose metabolism.
Key targets of the thyroid gland, which regulates metabolism, thermogenesis, and development:
- Thyroxine (T4)—the primary hormone produced by the thyroid which serves as a prohormone converted to T3 in peripheral tissues. It reflects overall thyroid output.
- Triiodothyronine (T3)—the biologically active thyroid hormone. It increases mitochondrial activity, oxygen consumption, thermogenesis, and lipid and carbohydrate metabolism.
- Thyroglobulin—a precursor protein for thyroid hormone synthesis which is used as a tumor marker in differentiated thyroid cancer.
- Thyroid peroxidase antibodies (TPO-Ab)—indicate autoimmune thyroid destruction, which leads to hypothyroidism and metabolic slowing.
Key targets of the pancreas (endocrine), which is central to glucose and energy metabolism:
- Insulin—the key anabolic hormone that promotes glucose uptake, glycogen synthesis, lipogenesis, and protein synthesis. Insulin deficiency or resistance underlies diabetes.
- C-Peptide—reflects endogenous insulin production and beta-cell function, useful in distinguishing between diabetes types.
- Glucagon—counteracts insulin by stimulating glycogen breakdown and gluconeogenesis during fasting.
- Amylin—slows gastric emptying and suppresses glucagon after meals, supporting postprandial glucose control.
- HbA1c—reflects average glucose exposure over ~3 months and integrates long-term metabolic control.
These glands are central hubs for hormone synthesis and release. However, modern research has repeatedly shown that hormone production is not exclusive to glands. Many other tissues and organs also produce hormones that regulate metabolism, appetite, immune function, growth, and more [4].
A particularly well-studied example comes from adipose tissue (body fat). Once considered a passive energy store, adipose tissue is now recognized as a biologically active endocrine organ. Fat cells (adipocytes) secrete hormones—often called adipokines—such as leptin and adiponectin, that circulate in the bloodstream and directly influence energy balance, appetite, insulin sensitivity, and metabolic health. [5]
- Leptin, produced in proportion to fat mass, acts on the hypothalamus to reduce food intake and modulate energy expenditure, linking nutritional state to neuroendocrine control of appetite.
- Adiponectin, also derived from adipose tissue, plays an important role in enhancing insulin sensitivity and protecting against inflammation and vascular dysfunction.
Other tissues similarly contribute to endocrine signals. For example, cells in the gastrointestinal tract release hormones like ghrelin that stimulate appetite and influence metabolic processes, while the kidneys, heart, skin, and placenta secrete hormones involved in blood pressure regulation, calcium balance, immune modulation, and pregnancy physiology.
Endocrinology and metabolism ELISA kits
Endocrinology and metabolism ELISA kits enable sensitive, quantitative measurement of hormones and metabolic biomarkers in serum, plasma, cell culture supernatants, and other sample types—supporting research in diabetes, obesity, thyroid function, stress biology, and related pathways.
For example, GLP-1 (glucagon-like peptide-1) is critically important for research because it sits at the intersection of glucose metabolism, appetite regulation, cardiovascular biology, and therapeutic innovation. It helps provide a unifying biological pathway linking type 2 diabetes, obesity, and cardiometabolic disease.
Linearity-of-dilution data and the standard curve of Human GLP-1 using the Human GLP-1 ELISA Kit are shown in Figure 3 and Figure 4.
Search endocrinology ELISA kits Search metabolic kits
Learn more about ELISA kits and components
Popular endocrine and metabolic protein targets and ELISA performance data
Table 1. View our kits for the listed popular targets.
Table 2. List of publications highlighting Invitrogen ELISA kits used in endocrinology and metabolic research.
| Reference | Publication title | Target/s | Testing method |
|---|---|---|---|
| Gurevich et al. 2020. | iPSC-derived hepatocytes generated from NASH donors provide a valuable platform for disease modeling and drug discovery | Albumin | Human Albumin ELISA Kit |
| Ma et al. 2022. | Sennoside A Induces GLP-1 Secretion Through Activation of the ERK1/2 Pathway in L-Cells | GLP-1 | Human GLP-1 ELISA Kit |
| Dargam et al. 2025. | Phosphate salt selection affects mortality and vascular calcification in the adenine-induced chronic kidney disease mouse model | Urea nitrogen (BUN) | Urea Nitrogen (BUN) Colorimetric Detection Kit |
| Chen et al. 2021. | Decreased blood vessel density and endothelial cell subset dynamics during ageing of the endocrine system | Thyroxine (T4) | Thyroxine (T4) Competitive ELISA Kit |
| Stratton et al. 2020. | Four Weeks of Time-Restricted Feeding Combined with Resistance Training Does Not Differentially Influence Measures of Body Composition, Muscle Performance, Resting Energy Expenditure, and Blood Biomarkers | Cortisol | Human Cortisol Competitive ELISA Kit |
Endocrinology and metabolism ProQuantum high sensitivity immunoassays
Invitrogen ProQuantum high-sensitivity immunoassays are designed to help provide a high-performance quantitative protein measurement from 2–5 µL of sample. The assay is fast and easy to run, with no proprietary instrument to purchase. Utilizing proximity-based amplification technology, ProQuantum assays offer the analyte specificity of high-affinity antibody-antigen binding with the signal detection and amplification capabilities of real-time PCR to achieve a highly sensitive protein quantitation assay. ProQuantum assays can typically detect lower levels of protein with lower sample consumption than traditional methods and offer a broader dynamic range. This is important to accurately detect variations in concentration of endocrine and metabolic markers as they can vary depending on the metabolic state and disease state, as well as time of day.
Find endocrinology-related ProQuantum assays
Learn more about how the ProQuantum immunoassays work
Read BioProbes Journal article: Introducing ProQuantum High-Sensitivity Immunoassays—The new generation of target-specific protein quantitation
Table 3. View our ProQuantum immunoassays for the following popular targets.
Endocrinology and metabolism ProcartaPlex multiplex immunoassays
Invitrogen ProcartaPlex multiplex immunoassays help provide a powerful platform for the simultaneous quantification of up to 9 endocrine and metabolic biomarkers, supporting researchers in illuminating the reasons for endocrine dysregulation, metabolic changes, and complex feedback mechanisms across multiple organ systems. ProcartaPlex assays demonstrate strong correlation to traditional ELISA methods, supporting confidence in data comparability while enabling multiplex efficiency (Figure 6).
Hormones can be present across a wide range of concentrations. ProcartaPlex assays are designed with a broad dynamic range, allowing accurate detection of these varying levels within a single run. This enables reliable measurement of hormones, verified in cerebrospinal fluid (CSF), serum, plasma, and cell culture supernatant samples. Other biological samples might be suitable for use in the assays.
As hormones can also influence immunological and neurological processes, the ProcartaPlex platform enables simultaneous measurement of hormones alongside key immune and neurological biomarkers in a single multiplex panel. Because many hormones, particularly steroid and small peptide hormones, are low-molecular-weight targets with limited epitopes, competitive immunoassays are the preferred format for accurate quantification. ProcartaPlex combines competitive assays for small-molecule hormones with sandwich assays for larger proteins such as cytokines and peptide biomarkers, all within the same workflow. This can be exploited by combining several ProcartaPlex Simplex kits into a panel or by custom blended Mix & Match panels. This integrated approach has been extensively tested for cross-compatibility and can provide significant efficiency in time, sample volume, and cost compared with running multiple single-plex assays.
Select from our preconfigured panels described in Table 4 or use the Panel Configurator below to customize your specific panel.
ProcartaPlex Panel Configurator
Learn more about ProcartaPlex multiplex immunoassays
Preconfigured endocrinology multiplex immunoassay panels and performance data

Figure 8A. Progesterone serum levels in females across physiological states. Serum samples from pregnant (n=8), non-pregnant (n=8), and post-menopause (n=4) females were run in duplicates using Invitrogen ProcartaPlex Human Steroid and Thyroid Hormones Panel, 7plex. Compared with age-matched healthy, non-pregnant controls, pregnant females exhibit significantly increased progesterone levels, whereas postmenopausal females show significantly reduced progesterone levels relative to healthy, non-pregnant controls.
Figure 8B. Testosterone serum levels in females and males. Serum samples from female (n=10) and male (n=10) individuals, including NIST® SRM® Standard Reference Material® 971a with a certified concentration value for males (5,838 ng/mL, indicated in orange), were run in duplicates using Invitrogen ProcartaPlex Human Steroid and Thyroid Hormones Panel, 7plex.
Both data are presented as medians with interquartile ranges (IQR). Group differences and statistical significance were assessed by the Mann–Whitney U test (two-tailed); * p<0.05, ** p<0.01, *** p<0.001, **** p<0.0001.
Table 4. Preconfigured ProcartaPlex multiplex immunoassay panels for endocrinology.
| Human multiplex hormone panels | ||
|---|---|---|
| Name | Size | Cat. No. |
| ProcartaPlex Human Neuropeptide and Pituitary Hormones Panel, 9plex Target list [bead region]: Neuropeptide Y [42], ACTH [67], Cholecystokinin (CCK) [19], Follicle Stimulating Hormone (FSH) [77], Growth Hormone (GH) [48], Thyroid Stimulating Hormone (TSH) [28], Luteinizing Hormone (LH) [53], Prolactin [22], hCG [56] | 96 tests | EPX090-15861-901 |
| ProcartaPlex Human Steroid- and Thyroid Hormones Panel, 7plex Target list [bead region]: Triiodothyronine (T3) Competitive [38], Thyroxine (T4) Competitive [63], Testosterone Competitive [37], Estradiol (E2) Competitive [26], Progesterone Competitive [35], DHEA-S Competitive [43], Cortisol Competitive [12] | 96 tests | EPX070-15862-901 |
Endocrinology and metabolism QuantiGene multiplex immunoassays
QuantiGene assays are highly sensitive and specific for the detection and quantification of RNA targets. This technology utilizes branched DNA signal amplification to enable the measurement of gene expression levels in a variety of sample types, including blood, tissue, and cell lysates. With up to 80 genes simultaneously measured in a single sample, researchers can focus on endocrinology and hormone markers implicated in biochemical processes. A few examples of published data using QuantiGene panels are highlighted in Table 5.
There are several advantages to bead-based assays over traditional methods of gene expression analysis. QuantiGene assays help provide a highly multiplexed approach, allowing for the simultaneous measurement of multiple targets in a single sample using the same Luminex xMAP technology as ProcartaPlex assays. This capability not only helps save time and resources but also helps provide a more comprehensive understanding of gene expression patterns. The assay is also highly sensitive, enabling the detection of low-abundance targets with high precision.
QuantiGene gene expression assays are versatile tools that can be customized to help meet your specific research needs. Researchers looking to study genes implicated in metabolism or obesity can choose from a selection of preconfigured panels in Table 6. Alternatively, by using the Panel Configurator below, you can choose from a large inventory of verified genes and pathways of interest to create a customized panel for studying endocrinology.
Learn more about QuantiGene multiplex immunoassays
Table 5. List of publications highlighting custom-designed QuantiGene assays for studying endocrinology.
| Reference | Publication summary |
|---|---|
| Ivanova et al., 2024. | A preliminary study was conducted using a custom QuantiGene panel, which was used to investigate the relationship between genetic expression markers in schizophrenic males with metabolic disorders. |
| Baruch, et al., 2023. | The group aimed to characterize Growth Differentiation Factor 15 (GDF15) as a hormone biomarker that is secreted within the central nervous system. A custom QuantiGene plex panel was designed to measure genes of interest in the ISR or Gdf-15 pathways. |
| Bellet et al., 2023. | To determine how much the tryptophan metabolism transcriptional pathway is regulated via the circadian system, mRNA levels of lung and ileum samples of mice were quantified using a custom-designed QuantiGene plex panel. |
| Serradas et al., 2021. | A custom QuantiGene panel was used to investigate the sweet taste transduction pathway and evaluate the hypothesized changes from metabolic diseases, including GLP-1. |
| Beckman et al., 2013. | To explore the endocrine growth axis of coho salmon, a custom QuantiGene plex panel was designed. Liver homogenates were prepared, and the mRNA levels of various hormones and receptors were measured. |
Table 6. Preconfigured QuantiGene gene expression multiplex immunoassay panels for metabolism and obesity research.
| Product Name | Size | Cat. No. |
|---|---|---|
| Human endocrinology panels | ||
| QuantiGene Plex Human Metabolism Panel, 25-plex Target list: PPARG, ADRB2, UCP1, INSR, IRS1, GNPDA2, TBC1D1, FABP4, TNNI3K, TFAP2B, PCSK1, MAF, TMEM160, CLOCK, ADIPOQ, HHEX, PLIN1, SLC6A14, FAIM2, SIRT1, GCG, IAPP, PPIB, TBP, HPRT1 | 1 plate | QGP-125-HUMETAB |
| QuantiGene Plex Human Obesity Panel, 20-plex Target list: MC4R, LEP, LEPR, GLP1-R, GCG (GLP-1), TMEM18, BDNF, KCTD15, SH2B1, MTCH2, NEGR1, SEC16B, FTO, POMC, MC3R, NMB, INHBE, PPIB, TBP, HPRT1 | 1 plate | QGP-120-HUOBESITY |
| Mouse endocrinology panels | ||
| QuantiGene Plex Mouse Metabolism Panel, 25-plex Target list: Pparg, Adrb2, Ucp1, Insr, Irs1, Gnpda2, Tbc1d1, Fabp4, Tnni3k, Tfap2b, Pcsk1, Maf, Tmem160, Clock, Adipoq, Hhex, Plin1, Slc6a14, Faim2, Sirt1, Gcg, Iapp, Tbp, Hprt1, Ppib | 1 plate | QGP-125-MSMETAB |
| QuantiGene Plex Mouse Obesity Panel, 20-plex Target list: Mc4r, Lep, Lepr, Glp1r, Gcg, Tmem18, Bdnf, Kctd15, Sh2b1, Mtch2, Negr1, Sec16b, Fto, Pomc, Mc3r, Nmb, INHBE, PPIB, TBP, HPRT1 | 1 plate | QGP-120-MSOBESITY |
Additional resources for endocrinology research
Competitive ELISA and other formats
Biomarker quantitation assay guide
ProcartaPlex Panel Configurator
免疫测定仪器
References
- Campbell, Miles. and Ishwarlal Jialal. “Physiology, Endocrine Hormones.” StatPearls, StatPearls Publishing, 26 September 2022.
- Marques-Deak, A., Cizza, G. & Sternberg, E. Brain-immune interactions and disease susceptibility. Mol Psychiatry10, 239–250 (2005).
- Vela-Patiño, Sandra et al. “Neuroendocrine-immune Interface: Interactions of Two Complex Systems in Health and Disease.” Archives of medical research vol. 53,3 (2022): 240–251.
- Liu, Chang, and Xiaojiao Li. “Role of leptin and adiponectin in immune response and inflammation.” International immunopharmacology vol. 161 (2025): 115082.
- Taylor, Erin B. “The complex role of adipokines in obesity, inflammation, and autoimmunity.” Clinical science (London, England : 1979) vol. 135,6 (2021): 731–752.
仅供科研使用,不可用于诊断目的。











