Search

table of Elements
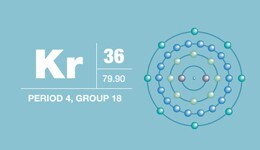
Krypton
Krypton X-ray photoelectron spectra, krypton electron configuration, and other elemental information.
Join the conversation
Krypton • Noble Gases
Primary XPS region: Kr3d
Overlapping regions: Au4f
Binding energies of common chemical states:
| Chemical state | Binding energy Kr3d3/2 |
|---|---|
| Implanted Kr | 88 eV |
Experimental information
N/A
Interpretation of XPS spectra
N/A
General comments
- Kr is a gas and is not commonly found in XPS spectra.
- Kr may be observed as implanted atoms, if a material has been subjected to Kr ion bombardment using Kr gas instead of the more usual Ar gas.
About this element
Symbol: Kr
Date of discovery: 1898
Name origin: Greek kryptos
Appearance: colorless
Discoverer: Sir William Ramsey
Obtained from: production of liquid air
Melting point: 115.95 K
Boiling point: 119.75 K
Density[kg/m3]: 3708
Molar volume: 27.99 × 10-6 m3/mol
Protons/Electrons: 36
Neutrons: 48
Shell structure: 2,8,18,8
Electron configuration: [Ar]3d104s24p6
Oxidation state: 0 (unknown)
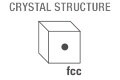
Crystal structure: cubic face centered
Krypton, named after the Greek word kryptos, meaning “hidden”, was discovered in 1898. The element was found in residue of the evaporated components of liquid air by W. Ramsay and M. Travers. The vibrant green and orange spectral-signatured element is considered a noble gas due to its low chemical reactivity. The element’s gas concentration in earth’s atmosphere is about 1 ppm and is isolated by fractioning liquid air. Krypton is most often combined with other rare gases to make fluorescent lamps. Historically, krypton was widely considered to be chemically inert; however, since the 1960’s, studies have found several compounds of krypton – including krypton diflouride and an oxyacid salt containing krypton.