Search

table of Elements
Hydrogen
Hydrogen X-ray photoelectron spectra, hydrogen electron configuration, and other elemental information.
Join the conversation
Hydrogen • Non-Metals
Primary XPS region: N/A
Overlapping regions: N/A
Binding energies of common chemical states: N/A
- Hydrogen in any compound does not have a core electron so it will not produce a core photoelectron peak
- Any signal would only appear in the valence band, but the photoelectron cross-section is also very low and so is never directly observed in XPS spectra.
- Reflected Electron Energy Loss Spectroscopy (REELS) is a possibility for detecting hydrogen [1].
N/A
- The presence of hydrogen may be inferred if the amount of other elements is greater than expected.
- E.g., excess oxygen on the surface of a metal oxide may indicate the presence of H in water or hydroxides.
- Hydrogen may also be inferred in the case of H-terminated Si.
- Almost no surface oxide is present in the Si2p spectrum.
- Metal hydrides tend to be highly reactive with oxygen or moisture, so they are likely to be observed as oxides or hydroxides after exposure to air.
Symbol: H
Date of discovery: 1766
Name origin: Greek hydro and gennan
Appearance: colorless
Discoverer: Henry Cavendish
Obtained from: mines, oil, gas wells
Melting point: 14.01 K
Boiling point: 20.28 K
Density[kg/m3]: 0.08988
Molar volume: 11.42 × 10-6 m3/mol
Protons/electrons: 1
Neutrons: 0
Shell structure: 1
Electron configuration: 1s1
Oxidation state: 1,-1
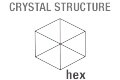
Crystal structure: hexagonal
Hydrogen is the most abundant element in the universe, making up 75% of matter by mass and over 90% by number of atoms. This element is found in great quantities in stars and gas giant planets. Lighter than air, hydrogen had been used as a lifting agent in balloons and airships. However, due to its risk of fire, its use has been discontinued following the Hindenburg disaster in 1937. Hydrogen isotopes are used in the production of hydrogen bombs. In the solar wind, hydrogen’s plasma interacts with the earth’s magnetosphere, giving rise to Birkeland currents and the aurora borealis.