Search

Materials Science
Ultraviolet Photoelectron Spectroscopy
Ultraviolet photoelectron spectroscopy explained – learn how it works and how it compares to X-ray photoelectron spectroscopy.
Ultraviolet photoelectron spectroscopy
Ultraviolet photoelectron spectroscopy operates on the same principles as XPS, the only difference being that ionizing radiation at energies of 10s of eV are used to induce the photoelectric effect, as opposed to photons of greater than 1 keV that are used in XPS. In the laboratory setting ultraviolet photons are produced using a gas discharge lamp, typically filled with helium, although other gases such as argon and neon can also be used. The photons emitted by helium gas have energies of 21.2 eV (He I) and 40.8 eV (He II).
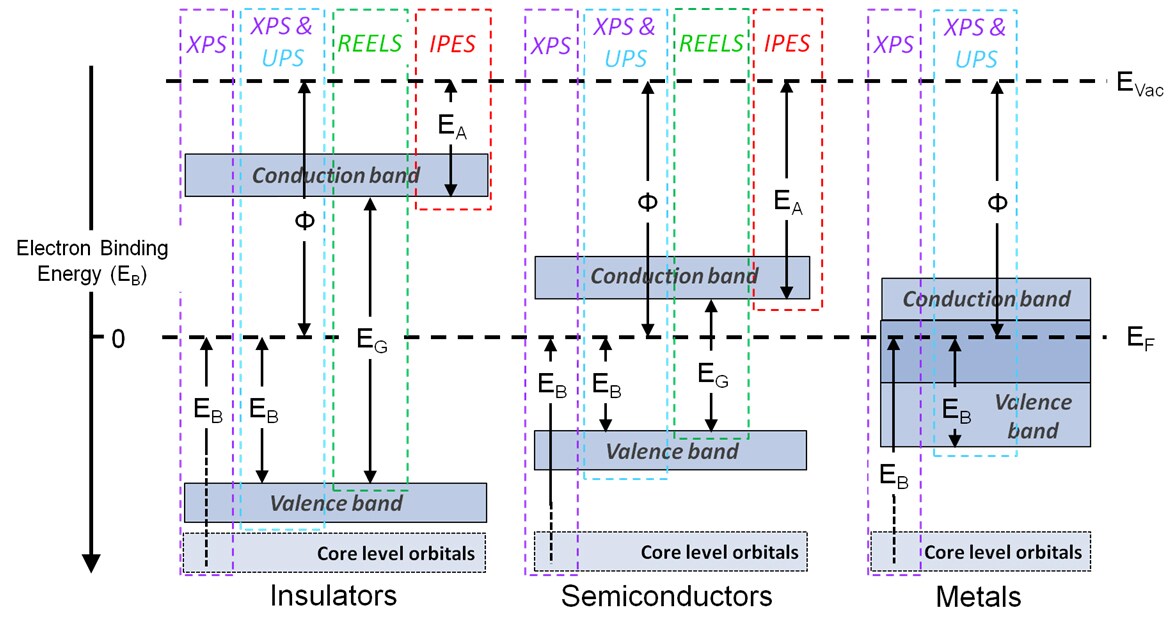
As lower energy photons are used, most core level photoemissions are not accessible using ultraviolet photoelectron spectroscopy, so spectral acquisition is limited to the valence band region. There are two types of experiment performed using ultraviolet photoelectron spectroscopy: valence band acquisition and electronic work function measurement.
Valence bands
Many of the molecular orbitals from which valence band photoelectron signal originates possess a high degree of hybridization, therefore the shifts in peak binding energy are far more varied and subtle than those observed for core level photoemission peaks. For this reason valence band spectra are predominantly used for material characterization through spectral fingerprinting, and individual peak assignment is either performed on surfaces with well-known electronic structure, or in conjunction with computational studies. Due to this ambiguity in the assignment of valence band peaks, these spectra are not used for quantification.
Ultraviolet photoelectron spectroscopy is also widely used to collect valence band spectra, the combination of both XPS and ultraviolet photoelectron spectroscopy to investigate the valence band can be extremely powerful as the ionization cross section of an orbital is dependent on the incident photon energy, therefore different electronic transitions and states can be probed by using different photon energies.
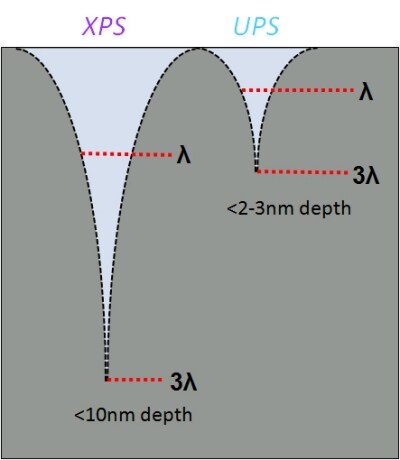
Ultraviolet photoelectron spectroscopy also exhibits greater surface sensitivity than XPS, the inherent surface sensitivity of XPS is due to the short inelastic mean free path (IMFP, or λ) of free electrons within a solid, with the so-called “information depth” from which > 99% of a photoemission signal originates conventionally being defined at 3 mean free path lengths from the surface, which in XPS is often quoted as 10 nm. This is an approximation as the IMFP of an electron is determined by the material properties of the solid media through which it is travelling and its kinetic energy, with electrons of lower kinetic energy having shorter path lengths. The lower incident photon energies used in ultraviolet photoelectron spectroscopy give emit photoelectrons of much lower kinetic energies than those measured in XPS, therefore giving ultraviolet photoelectron spectroscopy an approximate information depth of 2-3 nm.

Electronic work function
The difference between the Fermi level and Vacuum level is referred to as the electronic work function, a material property applied in the development of electronics devices, for example where matching of valence and conduction bands in multilayered devices is required. As a surface property, the work function is strongly influenced by variation in composition or structure at the surface, such as atmospheric contamination.
The electronic work function is acquired spectroscopically by measuring the difference between the Fermi Level and the cutoff of the ‘tail’ at the low kinetic energy end of the spectrum (a.k.a. spectrum width) and subtracting this value from the incident photon energy. This value can of course be measured using X-ray incident radiation; however, ultraviolet photoelectron spectroscopy allows work function calculation from a single spectrum.
When measuring the electronic work function using photoelectron spectroscopy it is necessary to apply a small bias (typically 5-10 V) to the sample surface, so as to deconvolute the true work function of the surface from the internal work function of the spectrometer.



