Search

table of Elements
Calcium
Calcium X-ray photoelectron spectra, calcium electron configuration, and other elemental information.
Join the conversation
Calcium • Alkaline Earth Metal
Primary XPS region: Ca2p
Overlapping regions: Mg KLL
Binding energies of common chemical states:
| Chemical state | Binding energy Ca2p3/2 |
|---|---|
| CaCO3 | 347.2 eV |
| Ca3(PO4)2 | 347.4 eV |
Charge referenced to adventitious C1s peak at 284.8eV.
Experimental information
- In the presence of a significant magnesium concentration, collect Ca2s as well as Ca2p region.
Interpretation of XPS spectra
- In the presence of magnesium, check for Ca2s to prevent misinterpreting Mg KLL as Ca2p.
- There is only a small range of Ca2p chemical shifts (<1eV) for typical compounds.
- Limited use in determining chemistry.
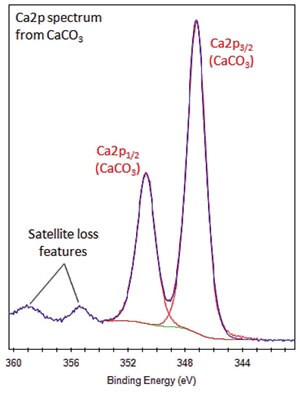
- Ca2p peak has clearly-spaced spin-orbit components (Δcarbonate=3.5eV).
- Satellite loss features are sometimes observed in the Ca2p region.
- Reasonably strong satellites are observed for CaCO3, for example, but only weak satellites for tribasic calcium phosphate.


General comments
- Since calcium is a highly reactive metal, it is always present as a compound.
- CaCO3 may degrade under prolonged X-ray exposure, giving CaO+CO2.
- Calcium compounds such as calcium oxide or hydroxide may react with atmospheric CO2, forming carbonates.
About this element
Symbol: Ca
Date of discovery: 1808
Name origin: Latin calcis
Appearance: silvery
Discoverer: Sir Humphrey Davy
Obtained from: chalk, limestone, marble
Melting point: 1112.15 K
Boiling point: 1757.15 K
Density[kg/m3]: 1550
Molar volume: 26.20 × 10-6 m3/mol
Protons/Electrons: 20
Neutrons: 20
Shell structure: 2,8,8,2
Electron configuration: [Ar]4s2
Oxidation state: 2
Crystal structure: cubic face centered
H. Davy first isolated calcium in 1808. It is the fifth most abundant element in the earth’s crust and is an important component for living organisms on the cellular level, as well as for the formation bones and teeth in higher organisms. Besides being an essential element for living things, calcium is used as a reducing agent in the extraction of thorium, zirconium, and uranium, and is an alloying agent in the production of aluminum alloys. Chemical refinery processes commonly use quicklime (CaO), which is also an important component in cement. Water-dissolving calcium compounds in the earth, such as limestone, form caves – including stalactites and stalagmites – and are the major cause of hard water.


