Search

table of Elements
Strontium
Strontium X-ray photoelectron spectra, strontium electron configuration, and other elemental information.
Join the conversation
Strontium • Alkaline Earth Metal
Primary XPS region: Sr3d
Overlapping regions: P2p
Binding energies of common chemical states:
| Chemical state | Binding energy Sr3d5/2 |
|---|---|
| SrO | 132.9 eV |
| SrCO3 | 133.4 eV |
| SrTiO3 | 133.1 eV |
Experimental information
- Sr3p1/2 overlaps C1s region.
Interpretation of XPS spectra
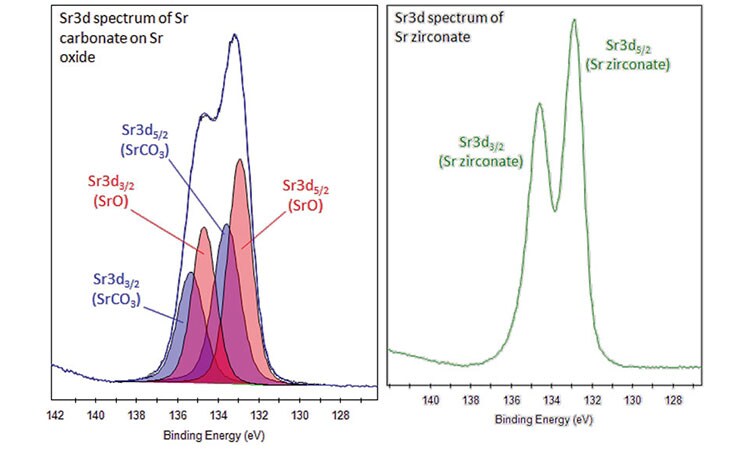
- Sr3d region has well-resolved spin-orbit components (Δ=1.76eV, ratio=0.690).
- When multiple chemical states are present, apparent resolution of these two spin-orbit components is reduced (compare Sr zirconate with SrO/SrCO3 spectra below).

General comments
- Sr metal reacts very readily with air to form its oxide, SrO.
- SrO reacts with atmospheric CO2 to make SrCO3.
- Strontium oxide is used in cathodic materials for solid oxide fuel cells.
About this element
Symbol: Sr
Date of discovery: 1790
Name origin: Strontian, Scotland
Appearance: silver-white
Discoverer: Adair Crawford
Obtained from: celestite, strontianite
Melting point: 1050 K
Boiling point: 1655 K
Density[kg/m3]: 2630
Molar volume: 33.94 × 10-6 m3/mol
Protons/Electrons: 38
Neutrons: 50
Shell structure: 2,8,18,8,2
Electron configuration: [Kr]5s2
Oxidation state: 2
Crystal structure: cubic
A soft, silvery element that tarnishes in air, strontium is commonly found in nature combined with other compounds, but never as the free element. Strontium reacts vigorously with water and its salts ignite spontaneously in air, imparting a brilliant crimson color often sought after in pyrotechnics. Strontium exists in three allotropic forms with at least 16 stable isotopes. With a half-life of 29 years, 90Sr is one of the best long-lived high-energy beta emitters known. It holds promise for applications in space vehicles and remote weather stations, in which a long-lived, lightweight, nuclear, electric power source is needed.


