Search

table of Elements
Aluminum
Aluminum X-ray photoelectron spectra, aluminum electron configuration, and other elemental information.
Join the conversation
Aluminum • Other Metal
Primary XPS region: Al2p
Overlapping regions: Pt4f, Cu3p
Binding energies of common chemical states:
| Chemical state | Binding energy Al2p |
|---|---|
| Al metal | 72.6 eV |
| Aluminosilicate | 74.4 eV |
| Al oxide | 74.6 eV |
| Al oxide on Al foil | 75.6 eV |
Oxide/foil referenced to Al2p metal peak.
Silicate and alumina referenced to adventitious C1s peak at 284.8eV.
Experimental Information
- In the presence of high platinum or copper concentrations, acquire Al2s as well as (or instead of) Al2p peak.
Interpretation of XPS Spectra
- Aluminum oxide is an insulating material and the oxide peak position may vary with the thickness of the film.
- The relative intensities of the oxide and metallic peaks will also change with oxide thickness.
- This effect allows XPS to measure the thickness of Al oxide films.
- The relative intensities of the oxide and metallic peaks will also change with oxide thickness.
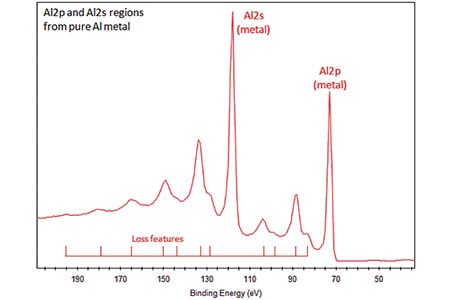
- Al2p peak has closely-spaced spin-orbit components (Δmetal=0.44eV).
- Splitting only observed for Al metal and under typical experimental conditions, manifests as asymmetry in peak rather than outright splitting.
- Splitting may be ignored for Al2p peaks from aluminum compounds, such as Al oxide.
- Metallic Al2p peak is ideally fit with asymmetric peak shapes.
- Compounds, such as Al oxide, have symmetric Al2p peaks.



- Aluminum metal exhibits energy loss features with a significant intensity.
- These features can overlap/obscure XPS peaks to higher binding energy, e.g., Si2p and Si2s peaks.
General comments
- Aluminum is a highly reactive metal, so always have a surface oxide present.
- The thickness of the oxide film may vary depending upon any previous cleaning treatment.
- Aluminum oxide may be hydrated with the presence of hydroxide species.
- These may react with atmospheric CO2 to form surface carbonates.
- After Ar+ sputtering, the reactive surface may adsorb oxygen and reform oxides.
Symbol: Al
Date of discovery: 1825
Name origin: Latin alumen
Appearance: silver
Discoverer: Hans Christian Ørsted
Obtained from: bauxite
Melting point: 933.52 K
Boiling point: 2740.15 K
Density[kg/m3]: 2700
Molar volume: 10.00 × 10-6 m3/mol
Protons/Electrons: 13
Neutrons: 14
Shell structure: 2,8,3
Electron configuration: [Ne]3s23p1
Oxidation state: 3
Crystal structure: cubic face centered
While ancient civilizations used aluminum salts for dyeing mordants and astringents, it wasn’t until 1825 that H. Ørsted isolated aluminum. The biggest breakthrough came in 1886, when C. Hall developed an inexpensive electrolytic process for extracting aluminum from the ore bauxite. This process is still commonly used today. Aluminum is valued for its light weight, strength, durability, and resistance to oxidation, and has propelled the use of aluminum alloys for aircraft and rockets. Evidence exists that high levels of aluminum may be toxic, although aluminum is generally considered less toxic than most heavy metals. Although the durability of aluminum is well-known, certain chemicals, such as mercury, can cause corrosion in aluminum. This is the reason why mercury thermometers are not allowed on aircrafts.


