Search

Magnetic Beads for Protein Purification |
Magnetic beads protein purification uses tiny, paramagnetic particles coupled with specific ligands that bind to target proteins. This technology is remarkable due to its efficiency, specificity, and ease of use. They are used to isolate and purify proteins from complex mixtures through magnetic separation, a faster alternative to centrifugation or filtration. Protein purification is essential in biotechnology and molecular biology, enabling researchers to isolate specific proteins for applications such as drug development and diagnostic testing. The most effective method of protein purification varies depending on the specific protein and application. Techniques such as affinity chromatography, ion exchange chromatography, and magnetic bead-based purification are commonly used.
This guide explores various protein purification techniques, with a focus on magnetic bead-based methods. We address key questions such as the advantages of magnetic beads, the most effective purification methods, and practical applications of these technologies.
Accelerate proteomics throughput (HTS) using magnetic separation
Speed and sample purity are major concerns for proteomics researchers, and strategies to achieve higher throughput for sample-handling and data-processing are desired. Magnetic particles are a solid support designed to be convenient for a variety of assays and procedures based on affinity purification. Paramagnetic beads are especially well-suited for automated procedures because instrumentation exists to easily mix, incubate, and separate the particles in 96-well plates without columns or centrifugation. Such ease of manipulation enables rapid processing of many samples and easy integration and compatibility with entire workflows.
Whether you’re isolating a recombinant fusion protein, enriching a target from lysate, or preparing samples for downstream analysis, the Thermo Scientific Magnetic Bead Technology Handbook shows how modern labs are moving beyond traditional chromatography to magnetic bead–based purification.
If you express, enrich, or isolate proteins, this handbook is for you.
Automated protein purification workflow
| Protein expression | Cell lysis and protein extraction | Purification with magnetic beads | Sample cleanup | Detection systems |
|---|---|---|---|---|
Automated magnetic protein purification selection guide
Magnetic beads for protein purification are highly effective. They help provide a rapid, efficient, and scalable method for isolating proteins with high specificity and purity, making them suitable for both manual and automated workflows.
| Magnetic beads (ng to μg binding capacity) | Magnetic agarose beads (mg capacity) | |
|---|---|---|
| IP-MS | Protein A/G Streptavidin | |
| IP/co-IP | Protein A/G Protein A Protein G | |
| Biotin IP | Streptavidin | |
| ChIP | Protein A/G Protein A Protein G | |
| Epitope tag | Anti-HA Anti-c-Myc | Glutathione Anti-DYKDDDDK |
| Antibody purification | Protein A/G Protein A Protein G Protein L | Protein A/G Protein A, alkali stable |
| IMAC | Ni-NTA | Ni-NTA Ni-IMAC (EDTA-compatible) |
| Surface reactive |
View non-magnetic affinity purification resin and kits
Kingfisher software protocols for automated protein purification
The table below includes Kingfisher software protocols for automated protein purification based on magnetic bead, affinity target, kit used, and binding strategy. Pierce magnetic beads are small, 1 μm particles that allow for high-throughput screening, purification, and immunoprecipitation with ng–μg protein yields. Alternatively, Pierce magnetic agarose beads consist of significantly larger, 10–40 μm particles and are used for high-capacity protein purification at mg–g scales. Both Pierce bead types can be automated on KingFisher instruments or used in a manual format with magnetic stands.
| Magnetic bead | Affinity target | Magnetic bead kits | Binding strategy | KingFisher software protocols |
|---|---|---|---|---|
| Anti-HA | HA-tagged fusion proteins | HA-Tag Magnetic IP/Co-IP kit | Direct | |
| Anti-c-Myc | Myc-tagged fusion proteins | Myc-Tag Magnetic IP/Co-IP kit | Direct | |
| Ni-NTA | His-tagged fusion proteins | Direct | ||
| NHS ester | Covalent immobilization of primary amine molecules (proteins, peptides, etc.) for affinity purification | Direct Magnetic IP kit | Direct | IP protocol Coupling protocol |
| Protein A/G | Antibodies (IgG containing Fc) | Classic Magnetic IP kit Crosslink Magnetic IP kit | Indirect | |
| Protein A | Antibodies (IgG containing Fc) | Indirect | ||
| Protein G | Antibodies (IgG containing Fc) | Indirect | ||
| Protein L | Antibodies (Ig containing kappa light chains) | Indirect | ||
| Streptavidin | Biotin or desthiobiotin | Indirect | Flex Instrument |
| Magnetic agarose bead | Affinity target | Binding strategy | KingFisher instrument protocols |
|---|---|---|---|
| Anti-DYKDDDDK | DYKDDDDK-tagged fusion proteins | Acid elution Peptide elution | |
| Protein A, Alkali Stable | mAbs and a subpopulation of scFv’s and Fab fragments containing a VH3 domain | ||
| Glutathione | Glutathione S-transferase (GST) fusion proteins | ||
| Ni-NTA | His-tagged fusion proteins | Direct | |
| Ni-IMAC | His-tagged fusion proteins, EDTA compatible | Direct (EDTA compatible chemistry) | |
| Protein A/G | Antibodies (IgG containing Fc) | Indirect |
Magnetic agarose beads consist of highly crosslinked agarose encapsulating a ferrimagnetic core. The beads are 10 to 40 µM in size and have higher binding capacity than traditional magnetic beads. We offer a variety of ligands for immunoprecipitation (IP), co-immunoprecipitation (co-IP), pull-down, and other high throughput affinity screening applications, utilizing immobilized Protein A/G, Ni-NTA, Ni-IMAC (EDTA Compatible), Glutathione, and Anti-FLAG. The beads are removed from the solution manually using a magnetic stand or by automation using an instrument such as the Thermo Scientific KingFisher Flex Magnetic Particle Processor. Automated instruments are especially useful for higher throughput purification and screening of purification conditions.
Choose the right protein purification strategy using magnetic agarose beads
| Pierce High-Capacity Protein A MagBeads, alkali stable | Pierce Protein A/G Magnetic Agarose Beads | Pierce Ni-NTA Magnetic Agarose Beads | Pierce High Capacity Ni-IMAC MagBeads, EDTA compatible | Pierce Glutathione Magnetic Agarose Beads | Pierce Anti-DYKDDDDK (FLAG) Magnetic Agarose Beads | |
|---|---|---|---|---|---|---|
| Binding target | Antibodies | Antibodies | His-tagged proteins | His-tagged proteins, EDTA compatible | GST-tagged proteins | FLAG-tagged proteins |
| Binding capacity | ≥40 mg/mL | ≥40 mg/mL | ≥70 mg/mL | 80 mg/mL | ≥12 mg/mL | ≥3 mg/mL |
| Order products | A53035 A53036 A53037 A53038 | 78609 | 78605 | A50588 | 78601 | A36797 |
| Magnetic bead | Affinity target | Magnetic bead kits | Binding strategy | KingFisher software protocols |
|---|---|---|---|---|
| Anti-HA | HA-tagged fusion proteins | HA-Tag Magnetic IP/Co-IP kit | Direct | |
| Anti-c-Myc | Myc-tagged fusion proteins | Myc-Tag Magnetic IP/Co-IP kit | Direct | |
| Ni-NTA | His-tagged fusion proteins | Direct | ||
| NHS ester | Covalent immobilization of primary amine molecules (proteins, peptides, etc.) for affinity purification | Direct Magnetic IP kit | Direct | IP protocol Coupling protocol |
| Protein A/G | Antibodies (IgG containing Fc) | Classic Magnetic IP kit Crosslink Magnetic IP kit | Indirect | |
| Protein A | Antibodies (IgG containing Fc) | Indirect | ||
| Protein G | Antibodies (IgG containing Fc) | Indirect | ||
| Protein L | Antibodies (Ig containing kappa light chains) | Indirect | ||
| Streptavidin | Biotin or desthiobiotin | Indirect | Flex Instrument |
| Magnetic agarose bead | Affinity target | Binding strategy | KingFisher instrument protocols |
|---|---|---|---|
| Anti-DYKDDDDK | DYKDDDDK-tagged fusion proteins | Acid elution Peptide elution | |
| Protein A, Alkali Stable | mAbs and a subpopulation of scFv’s and Fab fragments containing a VH3 domain | ||
| Glutathione | Glutathione S-transferase (GST) fusion proteins | ||
| Ni-NTA | His-tagged fusion proteins | Direct | |
| Ni-IMAC | His-tagged fusion proteins, EDTA compatible | Direct (EDTA compatible chemistry) | |
| Protein A/G | Antibodies (IgG containing Fc) | Indirect |
Magnetic agarose beads consist of highly crosslinked agarose encapsulating a ferrimagnetic core. The beads are 10 to 40 µM in size and have higher binding capacity than traditional magnetic beads. We offer a variety of ligands for immunoprecipitation (IP), co-immunoprecipitation (co-IP), pull-down, and other high throughput affinity screening applications, utilizing immobilized Protein A/G, Ni-NTA, Ni-IMAC (EDTA Compatible), Glutathione, and Anti-FLAG. The beads are removed from the solution manually using a magnetic stand or by automation using an instrument such as the Thermo Scientific KingFisher Flex Magnetic Particle Processor. Automated instruments are especially useful for higher throughput purification and screening of purification conditions.
Choose the right protein purification strategy using magnetic agarose beads
| Pierce High-Capacity Protein A MagBeads, alkali stable | Pierce Protein A/G Magnetic Agarose Beads | Pierce Ni-NTA Magnetic Agarose Beads | Pierce High Capacity Ni-IMAC MagBeads, EDTA compatible | Pierce Glutathione Magnetic Agarose Beads | Pierce Anti-DYKDDDDK (FLAG) Magnetic Agarose Beads | |
|---|---|---|---|---|---|---|
| Binding target | Antibodies | Antibodies | His-tagged proteins | His-tagged proteins, EDTA compatible | GST-tagged proteins | FLAG-tagged proteins |
| Binding capacity | ≥40 mg/mL | ≥40 mg/mL | ≥70 mg/mL | 80 mg/mL | ≥12 mg/mL | ≥3 mg/mL |
| Order products | A53035 A53036 A53037 A53038 | 78609 | 78605 | A50588 | 78601 | A36797 |
Achieve high-throughput sample processing and high protein binding capacity
On demand webinar: Learn how high-capacity magnetic agarose resin can be automated to achieve high throughput sample processing without sacrificing high protein binding capacity. Dr. Barbara Kaboord, Senior R&D Manager at Thermo Fisher Scientific, demonstrates the successful use of high-capacity magnetic agarose purification workflows for recombinant IgG antibody screening, and for the purification of recombinant proteins expressed in in vitro translation systems.
Figure 1. Comparison of protein purification results between Pierce Anti-DYKDDDDK magnetic agarose and another supplier. C-terminal DYKDDDDK-tagged Green Renilla Luciferase protein was expressed using the Thermo Scientific 1-Step Human High-Yield Maxi IVT Kit and immunoprecipitated using Pierce Anti-DYKDDDDK Magnetic Agarose or Sigma-Aldrich Anti-FLAG™ M2 Magnetic Beads using the KingFisher Flex Purification System. Tagged proteins were competitively eluted with Pierce 3x DYKDDDDK peptide and analyzed by western blot (A), Pierce Renilla Luciferase Glow Assay (B), and silver staining (C). Comparison of the starting lysate (L), elutions (E), and bead boiled samples (BB) show effective capture and elution of DYKDDDDK-tagged proteins with no background. Correlation of protein and activity levels indicate that a high level of Green Renilla luciferase activity is maintained after purification and competitive peptide elution.
Figure 2. Excellent performance of Pierce High-Capacity Ni-IMAC MagBeads, EDTA Compatible compared to magnetic beads from supplier C when purifying proteins from cell culture supernatant. Cell culture supernatant (Expi293) containing over-expressed His-tagged EPO (40 mg total protein) was applied to Pierce High-Capacity Ni-IMAC MagBeads, EDTA Compatible as well as Competitor C. Beads were processed using protocols with buffers recommended by the manufacturers. Binding was performed with all samples for 30 minutes. The beads were collected on a magnetic stand and the flow-through (unbound) fractions were saved for analysis. The beads were then washed twice, and bound protein was eluted with a 15-minute incubation in Elution Buffer. The eluates were resolved on an SDS-PAGE gel and stained with GelCode Blue Safe Protein Stain. Gel lanes were normalized by volume. M = MW marker, L = lysate load, FT = flow-through, W = wash, and E = elution.
*HC = High-capacity.
Figure 3. Exceptional capacity of Pierce High-Capacity Ni-IMAC MagBeads, EDTA Compatible compared to magnetic beads from Competitor C, on the Kingfisher Flex. Purified over-expressed 6xHis-GFP was applied to Pierce High-Capacity Ni-IMAC MagBeads, EDTA Compatible as well as Competitor C (0–100 mg GFP per 10 µL settled beads). Beads were processed using protocols with buffers recommended by the manufacturers on the Kingfisher Flex instrument. Binding was performed with all samples for 30 minutes. The beads were collected, and the flow-through (unbound) fractions were saved for analysis. The beads were then washed twice, and bound protein was eluted with a 15-minute incubation in Elution Buffer.
*HC = High-capacity
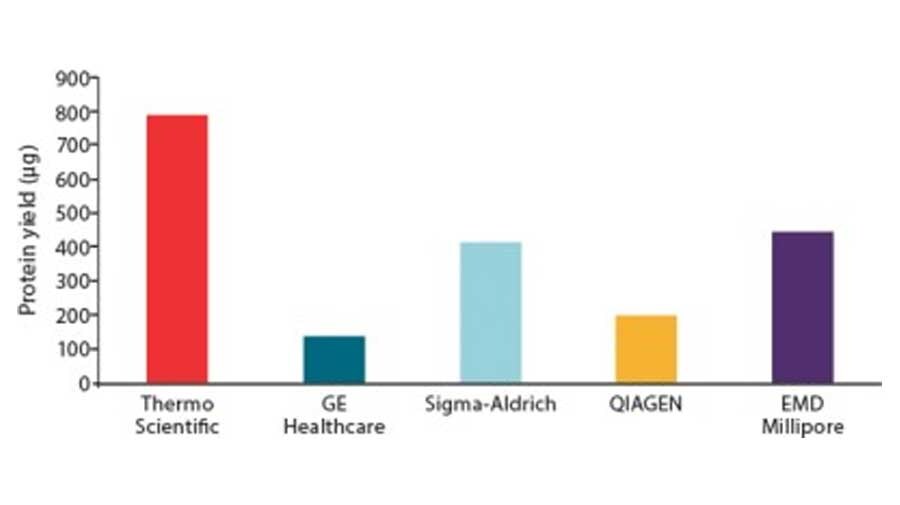

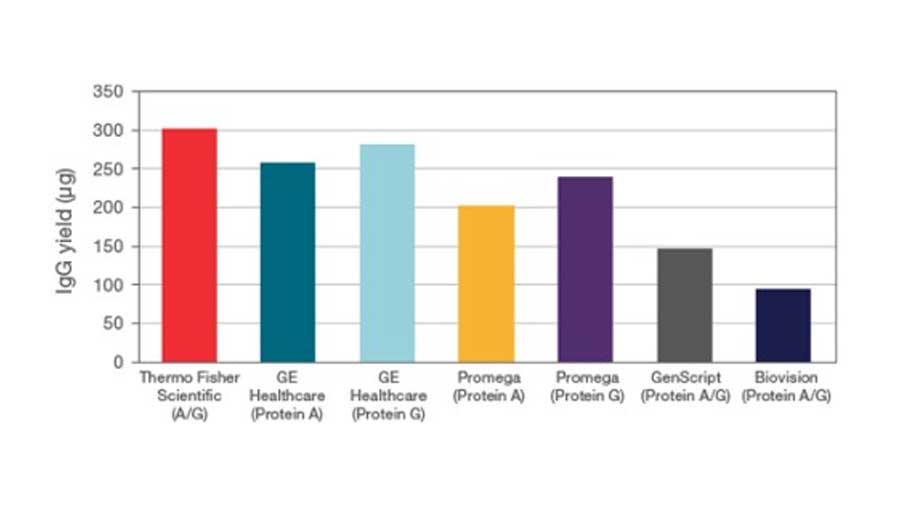
Figure 1. Comparison of protein purification results between Pierce Anti-DYKDDDDK magnetic agarose and another supplier. C-terminal DYKDDDDK-tagged Green Renilla Luciferase protein was expressed using the Thermo Scientific 1-Step Human High-Yield Maxi IVT Kit and immunoprecipitated using Pierce Anti-DYKDDDDK Magnetic Agarose or Sigma-Aldrich Anti-FLAG™ M2 Magnetic Beads using the KingFisher Flex Purification System. Tagged proteins were competitively eluted with Pierce 3x DYKDDDDK peptide and analyzed by western blot (A), Pierce Renilla Luciferase Glow Assay (B), and silver staining (C). Comparison of the starting lysate (L), elutions (E), and bead boiled samples (BB) show effective capture and elution of DYKDDDDK-tagged proteins with no background. Correlation of protein and activity levels indicate that a high level of Green Renilla luciferase activity is maintained after purification and competitive peptide elution.
Figure 2. Excellent performance of Pierce High-Capacity Ni-IMAC MagBeads, EDTA Compatible compared to magnetic beads from supplier C when purifying proteins from cell culture supernatant. Cell culture supernatant (Expi293) containing over-expressed His-tagged EPO (40 mg total protein) was applied to Pierce High-Capacity Ni-IMAC MagBeads, EDTA Compatible as well as Competitor C. Beads were processed using protocols with buffers recommended by the manufacturers. Binding was performed with all samples for 30 minutes. The beads were collected on a magnetic stand and the flow-through (unbound) fractions were saved for analysis. The beads were then washed twice, and bound protein was eluted with a 15-minute incubation in Elution Buffer. The eluates were resolved on an SDS-PAGE gel and stained with GelCode Blue Safe Protein Stain. Gel lanes were normalized by volume. M = MW marker, L = lysate load, FT = flow-through, W = wash, and E = elution.
*HC = High-capacity.
Figure 3. Exceptional capacity of Pierce High-Capacity Ni-IMAC MagBeads, EDTA Compatible compared to magnetic beads from Competitor C, on the Kingfisher Flex. Purified over-expressed 6xHis-GFP was applied to Pierce High-Capacity Ni-IMAC MagBeads, EDTA Compatible as well as Competitor C (0–100 mg GFP per 10 µL settled beads). Beads were processed using protocols with buffers recommended by the manufacturers on the Kingfisher Flex instrument. Binding was performed with all samples for 30 minutes. The beads were collected, and the flow-through (unbound) fractions were saved for analysis. The beads were then washed twice, and bound protein was eluted with a 15-minute incubation in Elution Buffer.
*HC = High-capacity



KingFisher magnetic particle processors
Prepare samples efficiently from a variety of materials with Thermo Scientific KingFisher Purification Systems, which offer highly versatile, automated magnetic-particle processing for DNA/RNA, protein, or cell purification from virtually any source. Using innovative magnetic particle separation technology, these systems help provide excellent reproducibility and quality. Thermo Scientific BindIt Software helps provides integrated or remote control of KingFisher instruments. Ready-to-use BindIt Software protocols (.bdz files) are available for a variety of applications with Pierce magnetic beads.
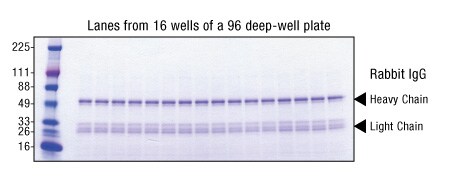
Figure 8. Consistency of Pierce High-Capacity Ni-IMAC MagBeads, EDTA Compatible purifications performed on the KingFisher Flex instument. Cell culture supernatant (Expi293) containing over-expressed His-tagged HSA (50 mg total protein) was applied to Pierce High-Capacity Ni-IMAC MagBeads, EDTA-Compatible (10 µL settled beads) in a 96-well plate. Beads were processed using the KingFisher Flex instrument. Binding was performed with all samples for 30 minutes. The beads were then washed twice, and bound protein was eluted with a 15-minute incubation in Elution Buffer. The eluates were resolved on an SDS-PAGE gel and stained with GelCode Blue Safe Protein Stain. Gel lanes were normalized to equivalent volume.
M = MW marker, L = lysate load, 1-12 = corresponding elution from plate row. CV’s <10%

Easily search and download automated protocols for Pierce magnetic agarose beads using BindIt PC software included with all KingFisher automated purification systems.
KingFisher purification instruments
Automated isolation, extraction, and purification of DNA, RNA, proteins, and cells. Applied Biosystems MagMAX
optimized kits and reagents provide easy-to-use protocols that remove manual steps and help save time.
KingFisher sample preparation systems for automated magnetic protein purification
 | KingFisher Apex SystemThe KingFisher Apex System is similar in specifications to the KingFisher Flex System, but redesigned with additional capabilities for peak performance, adaptability, and usability such as a barcode scanner, a cooling block, and a UV decontamination lamp. |
 | KingFisher Duo Prime SystemThe KingFisher Duo Prime System (low/medium throughput), meant for small labs and individual researchers, processes up to 12 samples per run. |
 | KingFisher Flex SystemThe KingFisher Flex System (medium/high throughput), the standalone laboratory workhorse, processes up to 96 samples per run and is excellent for high-throughput applications and processes 24 and 96 samples per run. |
Learning resources
For Research Use Only. Not for use in diagnostic procedures.




