Search

table of Elements
Titanium
Titanium X-ray photoelectron spectra, titanium electron configuration, and other elemental information.
Titanium • Transition Metal
Primary XPS region: Ti2p
Overlapping regions: Ru3p3/2, In3d3/2
Binding energies of common chemical states:
| Chemical state | Binding energy Tip3/2 |
|---|---|
| Ti metal | 454.1 eV |
| TiN | 454.9 eV |
| TiO2 | 458.5 eV |
| SrTiO3 | 458.4 eV |
Experimental information
N/A
Interpretation of XPS spectra
- Ti metal gives asymmetric Ti2p peak shapes, TiO2 has symmetric peaks shapes, and TiN has a complex peak shape, involving satellite features.
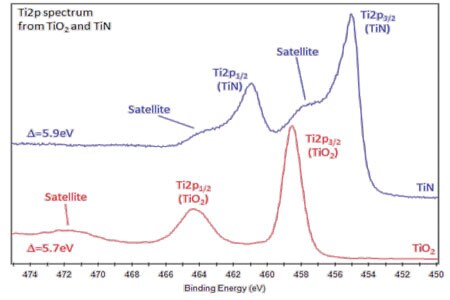
- Ti2p peak has significantly-split spin-orbit components (Δmetal=6.1eV).
- Splitting -value varies with chemical state (Δnitride=6.0eV, Δoxide=5.7eV).
- Typically FWHM for each spin-orbit component is the same, but for Ti2p, the Ti2p1/2 component is much broader than the Ti2p3/2 peak. Consequently, Ti2p1/2 peak is much shorter than expected.
- Caused by Coster-Kronig effect (post-ionization, Ti2p1/2 state is very short lived compared to Ti2p3/2 state).
- Causes difficulty in accurately peak fitting Ti2p region with multiple chemical states.

General comments
- Metal is readily oxidized.
- Titanium is used as a getter material for oxygen.
- TiO2 is readily reduced by argon monomer sputtering.
- Sub-oxides and/or metal may be formed during sputtering of the oxide.


About the element
Symbol: Ti
Date of discovery: 1791
Name origin: Greek Titans
Appearance: silverish
Discoverer: William Gregor
Obtained from: ilmenite, rutile
Melting point: 1941 K
Boiling point: 3560 K
Density[kg/m3]: 4507
Molar volume: 10.64 × 10-6 m3/mol
Protons/Electrons: 22
Neutrons: 26
Shell structure: 2,8,10,2
Electron configuration: [Ar]3d24s2
Oxidation state: 4
Crystal structure: Hexagonal
Titanium has a very high strength-to-weight ratio and is corrosion-resistant. Most titanium is used in the form of titanium dioxide (TiO2). Its silvery metallic color makes it very useful in paints, paper and plastics. Paint made with TiO2 reflects infrared radiation, making it useful in sunscreen and to astronomers. Because titanium is physiologically inert, it is used in joint replacement implants and is popular in body piercing. Titanium is not found unbound in nature and it occurs mainly in minerals. Rocks brought back from the moon contained about 12% TiO2.



